What type of bond is magnesium bromide?
.
Similarly, you may ask, what type of compound is magnesium bromide?
Magnesium bromide (MgBr2) is a chemical compound of magnesium and bromine that is white and deliquescent. It is often used as a mild sedative and as an anticonvulsant for treatment of nervous disorders.
One may also ask, is magnesium bromide a base? Predict whether aqueous solutions of the following salts will be acidic, basic, or neutral.
Salts Debriefing.
| Ammonium sulfide, (NH4)2S | acidic basic neutral |
|---|---|
| Ethyl ammonium chloride, CH3CH2NH3Cl | acidic basic neutral |
| Magnesium bromide, MgBr2 | acidic basic neutral |
People also ask, is magnesium bromide ionic?
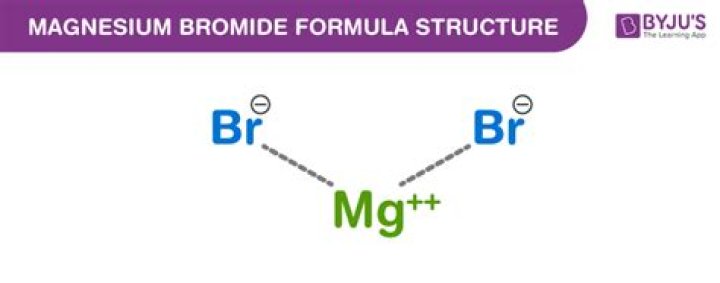
Ionic Compounds. Therefore, for the resulting compound to be neutral, two bromine anions must combine with one magnesium cation to form magnesium bromide (MgBr2).
Is magnesium bromide a metal or nonmetal?
Explanation: It is neither a metal nor a nonmetal. Magnesium bromide, MgBr2 , is an ionic compound containing the metal magnesium (Mg) and the nonmetal bromine (Br).
Related Question AnswersHow do you name MgBr2?
Answer and Explanation: The name for the compound MgBr2 is magnesium bromide. Mg is the symbol for magnesium, and Br is the symbol for bromine.What does magnesium bromide and chlorine yield?
The balanced equation for the reaction of magnesium bromide and chlorine is MgBr2 + Cl2 = Br2 + MgCl2. MgBr2 is the formula for magnesium bromide, Cl2 is the formula for chlorine gas, Br2 is the formula for bromine gas, and MgCl2 is the formula for magnesium chloride.How many atoms are in magnesium bromide?
Magnesium bromide (MgBr2)| PubChem CID: | 522691 |
|---|---|
| Structure: | Find Similar Structures |
| Molecular Formula: | Br2Mg |
| Synonyms: | Magnesium bromide (MgBr2) MgBr2 Magnesium bromide, 98% DTXSID7064865 EINECS 232-170-9 More |
| Molecular Weight: | 184.11 g/mol |
What is the charge of magnesium bromide?
As an ion it has a charge of 1-, Br-because it has to gain an electron to attain stable electronic configuration and octet valance shell structure resembling that of noble gases. So now we have got Mg2+ and Br-. To balance the chemical formula, there should be 2 bromide ions and one magnesium ion.Is magnesium bromide aqueous?
About Magnesium Bromide Most metal bromide compounds are water soluble for uses in water treatment, chemical analysis and in ultra high purity for certain crystal growth applications. Bromide in an aqueous solution can be detected by adding carbon disulfide (CS2) and chlorine.Why is magnesium bromide MgBr2?
? Magnesium bromide (MgBr2) is a chemical compound of magnesium and bromine that is white and deliquescent. The oxygen side will be directed towards the magnesium ions because the magnesium ions have a positive charge that will attract the negatively charged side of the water molecule.What percent of magnesium bromide is magnesium?
Percent composition by element| Element | Symbol | Mass Percent |
|---|---|---|
| Magnesium | Mg | 13.201% |
| Bromine | Br | 86.799% |
What are the formulas for ionic compounds?
Examples of Ionic Compounds| Compound Name | Formula | Cation |
|---|---|---|
| lithium fluoride | LiF | Li+ |
| sodium chloride | NaCl | Na+ |
| calcium chloride | CaCl2 | Ca2+ |
| iron(II) oxide | FeO | Fe2+ |



