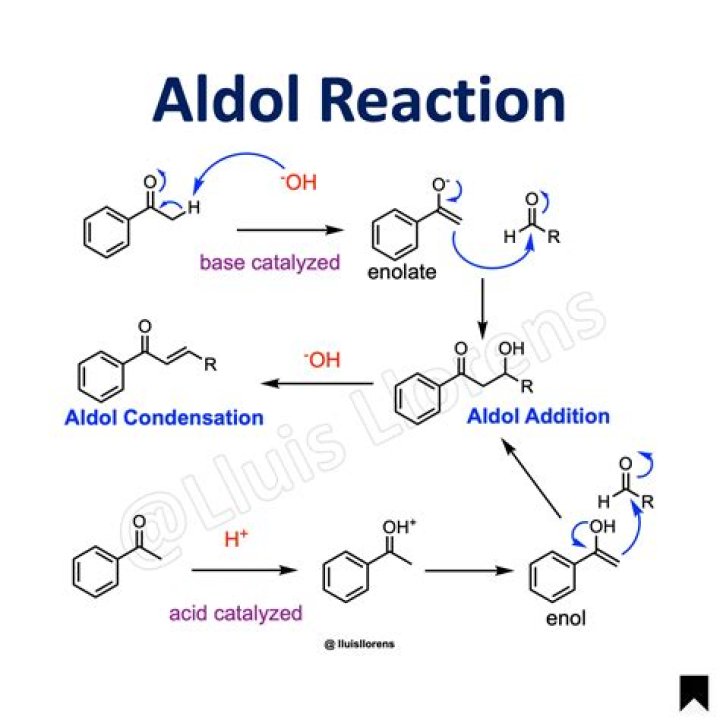
Aldol reactions are excellent methods for the synthesis of many enones or beta hydroxy carbonyls. Because of this, being able to predict when an aldol reaction might be used in a synthesis in an important skill..
Also asked, why are aldol reactions important?
Aldol condensations are important in organic synthesis, because they provide a good way to form carbon–carbon bonds. The reaction between an aldehyde/ketone and an aromatic carbonyl compound lacking an alpha-hydrogen (cross aldol condensation) is called the Claisen-Schmidt condensation.
what reaction type is an aldol reaction? Aldol condensation. An aldol condensation is a condensation reaction in organic chemistry in which an enol or an enolate ion reacts with a carbonyl compound to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), followed by dehydration to give a conjugated enone.
Considering this, what is an aldol product?
'Aldol' is an abbreviation of aldehyde and alcohol. When the enolate of an aldehyde or a ketone reacts at the α-carbon with the carbonyl of another molecule under basic or acidic conditions to obtain β-hydroxy aldehyde or ketone, this reaction is called Aldol Reaction.
What are Aldols used for?
It is also used in the manufacture of pharmaceuticals, such as a, b unsaturated ketones and the aromatic ketones known as chalcones. It is also generally used to create plasticizers. Aldol Condensation is a classic reaction.
Related Question Answers
What is the first step in an aldol reaction?
Aldol reaction: its chemistry and mechanism. Typically, aldehydes and ketones undergo an aldol reaction. This reaction involves 3 basic steps: Step 1: Electrophilic attack of a base at the alpha-carbon (C α?start subscript, α, end subscript) of the aldehyde or ketone to generate an “enolate” carbanion.What is double aldol condensation?
In Deep Discussion: Double Aldol Condensation. This reaction is called the aldol condensation. In every case the product results from the addition of one molecule of aldehyde (or ketone) to a second molecule in such a way that the α-carbon of the first becomes attached to the carbonyl carbon of the second.What is aldol condensation explain with example?
Aldol Condensation can be defined as an organic reaction in which enolate ion reacts with a carbonyl compound to form β-hydroxy ketone or β-hydroxy aldehyde, followed by dehydration to give a conjugated enone. Aldol Condensation plays a vital role in organic synthesis, creating a path to form carbon-carbon bonds.What is aldol condensation with example?
Aldol condensation is a reaction that results in the formation of a carbon-carbon bond when an enol (or enolate) molecule reacts with an aldehyde (or ketone). An example of the enol and enolate form of acetone is shown below.Who discovered the aldol reaction?
Alexander Borodin
What is Enolate?
An enolate ion is the anion formed when an alpha hydrogen in the molecule of an aldehyde or a ketone is removed as a hydrogen ion.Why is NaOH used in aldol condensation?
NaOH it undergoes self condensation as it contains alpha-hydrogen atom in its compound forming β-hydroxyaldehyde (an aldol) namely 3-Hydroxy butanal. This compound upon further heating will eliminate a molecule of water forming aldol condensation product namely Crotonaldehyde Or But-2-en-al.How do you stop aldol condensation?
Therefore, chemists have adopted many ways to prevent this from occurring when performing a crossed aldol reaction. - The use of a more reactive electrophile, and a non-enolizable partner.
- Making enolate ion quantitatively.
- Silyl enol ether formation.
How is aldol formed?
When the enolate of an aldehyde or a ketone reacts at the α-carbon with the carbonyl of another molecule under basic or acidic conditions to obtain β-hydroxy aldehyde or ketone, this reaction is called Aldol Reaction.Are aldol reactions reversible?
The Aldol Reaction. In the presence of acid or base catalysts the aldol reaction is reversible, and the beta-hydroxy carbonyl products may revert to the initial aldehyde or ketone reactants. In the absence of such catalysts these aldol products are perfectly stable and isolable compounds.What is self aldol condensation?
Self condensation: A condensation reaction in which two of the same molecule are condensed. This aldol condensation is a self condensation reaction because two molecules of acetone combine to form the product.What happens when aldol is heated?
Aldol reaction is an important organic reaction of aldehydes and ketones. The Aldol or the Ketol thus formed in the reaction upon heating eliminates a molecule of water (i.e. it undergoes dehydration) forming alpha, β-unsaturated Aldehydes or Ketones respectively.What is Haloform test?
The haloform reaction is the reaction of a methyl ketone with chlorine, bromine, or iodine in the presence of hydroxide ions to give a carboxylate ion and a haloform. There is one aldehyde that undergoes the haloform reaction, which is acetaldehyde.Do aldehydes react with NaOH?
Reagents : commonly a base such as NaOH or KOH is added to the aldehyde. The reaction involves an aldehyde enolate reacting with another molecule of the aldehyde. The products of these reactions are β-hydroxyaldehydes or aldehyde-alcohols = aldols. The simplest aldol reaction is the condensation of ethanal.Why do aldol products undergo dehydration?
Recall that ketones are less reactive than aldehydes towards nucleophilic addition due to steric and electronic effects. These Aldol products can often undergo dehydration (loss of water) to give conjugated systems (an elimination reaction) (overall = an aldol condensation).What is alpha hydrogen in aldol condensation?
Aldol Condensation Mechanism & Alpha Hydrogen Reactions. Aldol Condensation, in organic chemistry, is a condensation reaction of enol aka enolate and carbonyl compound. And Alpha-hydrogen atom is an atom in which the hydrogen atom is reacted with the alpha-carbon atom.What is the difference between aldol addition and aldol condensation reaction?
The aldol reaction is the first step in the aldol condensation. In the aldol reaction, an enolate nucleophile adds to an aldehyde or ketone to form a -hydroxy carbonyl compound, called an "aldol." That compound can then undergo elimination to form an ,-unsaturated carbonyl compound.What is the difference between aldol condensation and Cannizzaro reaction?
The main difference between aldol condensation and Cannizzaro reaction is that aldol condensation is a coupling reaction, whereas Cannizzaro reaction is a redox reaction.What is a crossed aldol reaction?
A crossed aldol reaction. In a crossed aldol reaction, two different carbonyl compounds are applied. If both carbonyl compounds contain an α hydrogen atom, both may act as electrophilic carbonyl compound, as well as nucleophilic enol or enolate. Benzaldehyde, which contains no α hydrogen atoms, is such an aldehyde.