What is the difference between competitive inhibition and noncompetitive inhibition?
.
Moreover, what is competitive and non competitive inhibition?
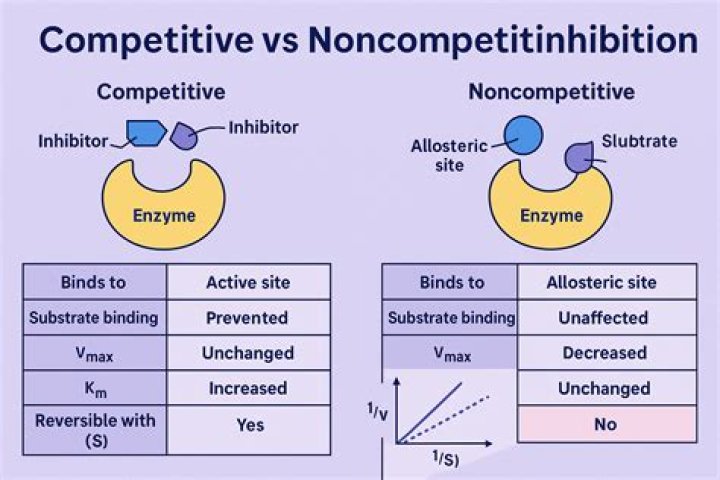
In competitive inhibition, an inhibitor molecule is similar enough to a substrate that it can bind to the enzyme's active site to stop it from binding to the substrate. In noncompetitive inhibition, an inhibitor molecule binds to the enzyme at a location other than the active site (an allosteric site).
Furthermore, how can you tell if an inhibitor is competitive or noncompetitive? Competitive vs. noncompetitive
- If an inhibitor is competitive, it will decrease reaction rate when there's not much substrate, but can be "out-competed" by lots of substrate.
- If an inhibitor is noncompetitive, the enzyme-catalyzed reaction will never reach its normal maximum rate even with a lot of substrate.
Additionally, what is the difference between non competitive inhibition and allosteric inhibition?
A noncompetitive inhibitor inhibits the action of an enzyme by binding to the enzyme somewhere other than the active site. An allosteric inhibitor binds to the enzyme, inducing it to assume an inactive form.
Is end product inhibition competitive or noncompetitive?
Often, the product of the last reaction in the pathway inhibits the enzyme that catalyses the first reaction of the pathway. This is called end-product inhibition and it involves non-competitive inhibitors.
Related Question AnswersWhat is an example of competitive inhibition?
In competitive inhibition, an inhibitor that resembles the normal substrate binds to the enzyme, usually at the active site, and prevents the substrate from binding. For example, methotrexate is a chemotherapy drug that acts as a competitive inhibitor.What are the 3 types of enzyme inhibitors?
There are three kinds of reversible inhibitors: competitive, noncompetitive/mixed, and uncompetitive inhibitors. Competitive inhibitors, as the name suggests, compete with substrates to bind to the enzyme at the same time. The inhibitor has an affinity for the active site of an enzyme where the substrate also binds to.What is another name for noncompetitive inhibition?
An inhibitor that is a chemical. Blocks/ binds the active site so the substrate cannot fit in. Describe non competitive inhibition? What's another name for this? Also called allosteric inhibiton.Why is competitive inhibition important?
Competitive Inhibitors Because of the presence of the inhibitor, fewer active sites are available to act on the substrate. But since the enzyme's overall structure is unaffected by the inhibitor, it is still able to catalyze the reaction on substrate molecules that do bind to an active site.What is competitive inhibition in biology?
' When a fake substrate binds to the active site of an enzyme, it can't be processed in the same way and it won't turn into a product. A fake substrate is called a competitive inhibitor. Competitive inhibitors bind the active site of an enzyme, preventing a real substrate from binding and a product from being formed.What is an example of a noncompetitive inhibitor?
In noncompetitive inhibition, a molecule binds to an enzyme somewhere other than the active site. For example, the amino acid alanine noncompetitively inhibits the enzyme pyruvate kinase. Alanine is one product of a series of enzyme-catalyzed reactions, the first step of which is catalyzed by pyruvate kinase.Is non competitive inhibition permanent?
Many Non-competitive Inhibitors are irreversible and permanent, and effectively denature the enzymes which they inhibit. However, there are a lot of non-permanent and reversible Non-competitive Inhibitors which are vital in controlling Metabolic functions in organisms.What does allosteric inhibition mean?
Explanation: An allosteric inhibitor by binding to allosteric site alters the protein conformation in active site of enzyme which consequently changes the shape of active site. Thus enzyme no longer remains able to bind to its specific substrate. This process is called allosteric inhibition.Why is allosteric inhibition important?
Allosteric regulation is very important, because it controls how much an enzyme will work. If the organism wants to shut down an enzyme, it will release an inhibitor, and if it wants to stimulate the enzyme into higher production, then it will release an activator.Is allosteric inhibition irreversible?
These types of inhibitors are called irreversible. However, other chemicals can transiently bind to an enzyme. These are called reversible. Reversible inhibitors either bind to an active site (competitive inhibitors), or to another site on the enzyme (non-competitive inhibitors).Is non competitive inhibition irreversible?
Non competitive inhibitors are usually reversible, but are not influenced by concentrations of the substrate as is the case for a reversible competive inhibitor. Irreversible Inhibitors form strong covalent bonds with an enzyme. These inhibitors may act at, near, or remote from the active site.How does pH affect enzyme activity?
pH: Each enzyme has an optimum pH range. Changing the pH outside of this range will slow enzyme activity. Extreme pH values can cause enzymes to denature. Enzyme concentration: Increasing enzyme concentration will speed up the reaction, as long as there is substrate available to bind to.Does Vmax change with enzyme concentration?
Chemical kinetics in general states that the reaction rate depends on the concentrations of the reactants. Although enzymes are catalysts, Vmax does depend on the enzyme concentration, because it is just a rate, mol/sec - more enzyme will convert more substrate moles into product.Why does non competitive inhibition lower Vmax?
In competitive inhibition, the Vmax does not change because increasing amounts of substrate can swamp the inhibitor (present in fixed concentration), allowing the enzyme to effectively not see the inhibitor at high substrate concentrations. In non-competitive inhibition, the Km does not change.What is Vmax enzyme?
Vmax is the reaction rate when the enzyme is fully saturated by substrate, indicating that all the binding sites are being constantly reoccupied.How do you plot a graph Michaelis Menten?
Plotting the Michaelis-Menten Curve Label the x-axis mM of [S] or concentration of substrate. Label the y ax- sec/micro-mole of V or velocity of reaction. Insert different values of [S] into the Michaelis-Menten equation, along with the values found for Km and Vmax, to solve for V.What are two ways to activate enzymes?
These include pH and temperature (discussed in the active site article), as well as:- Regulatory molecules. Enzyme activity may be turned "up" or "down" by activator and inhibitor molecules that bind specifically to the enzyme.
- Cofactors.
- Compartmentalization.
- Feedback inhibition.