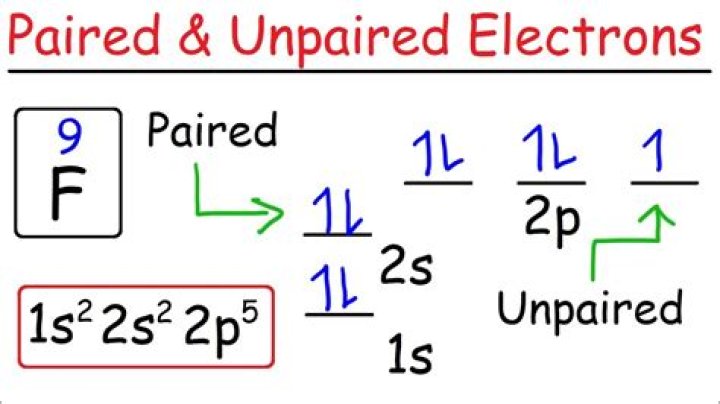
Five unpaired electrons
.
Simply so, how many unpaired electrons does MN have?
There are 5 unpaired electrons in 3d sub level.
Secondly, is MN paramagnetic or diamagnetic? The manganese(IV) oxide is the second most strongly attracted compound, and is paramagnetic. The permanganate shows no measurable attraction to the magnet, and appears to be diamagnetic. To theoretically determine the paramagnetism of the compound, we must determine the electron configurations.
Similarly, how many unpaired electrons does ND have?
The neodymium atom also can have a large magnetic dipole moment because it has 4 unpaired electrons in its electron structure as opposed to (on average) 3 in iron.
What is the electron configuration of ca2+?
The electron configuration of a Ca2+ ion is: 1s2 2s2 2p6 3s2 3p6, which is isoelectronic with the noble gas argon.
Related Question Answers
How many d electrons are in the valence shell of the Mn 3 cation how many unpaired electron spins?
Mn has oxidation number +3, so Mn3+ has electrong configuration [Ar]3d4. There are four unpaired electrons. Therefore, this octahedral complex has more unpaired electrons than the tetrahedral complex.What does unpaired electrons mean?
In chemistry, an unpaired electron is an electron that occupies an orbital of an atom singly, rather than as part of an electron pair. Each atomic orbital of an atom (specified by the three quantum numbers n, l and m) has a capacity to contain two electrons (electron pair) with opposite spins.How do you find unpaired electrons in ko2?
K O 2 KO_2 KO2 is potassium superoxide. It contains one unpaired electron due to the presence of superoxide ion ( O 2 − i o n ) (O^-_2, ion) (O2−ion) in it, which is paramagnetic. Whereas in A l O 2 − , B a O 2 AlO^-_2,, BaO_2 AlO2−,BaO2 and N O 2 + NO^+_2 NO2+, all the electrons are paired.How many unpaired electrons does arsenic have?
On the basis of pairs and unpaired electrons, an orbital diagram is formed. In case of Arsenic there are 3 unpaired boxes of electrons of 4p3 and rest 30 electrons in the pairs.Why is neodymium magnetic?
Neodymium is a rare-earth metal, and it is also ferromagnetic. It means; that like iron, neodymium can be magnetized. It is so strong because it has a very high state of saturation, magnetization. So, the magnetic energy the combination of neodymium, iron, and boron can store, makes the alloy very magnetic.Why is neodymium so magnetic?
Neodymium magnets are this strong because they have a very high level of magnetic saturation that allows them to create a large magnetic field. They are also very resistant to diamagnetism that they only lose their magnetic property when they are heated above their Curie temperature.Which magnets are the strongest?
The strongest permanent magnets in the world are neodymium (Nd) magnets, they are made from magnetic material made from an alloy of neodymium, iron and boron to form the Nd2Fe14B structure.Is neodymium harmful to humans?
The amount of neodymium in humans is quite small and, although the metal has no biological role, it can be effects on parts of the body: neodymium dust and salts are very irritating to the eyes. Ingested neodymium salts are regarded as only slightly toxic if they are soluble and non toxic if they are insoluble.Are powerful magnets dangerous?
The strong magnetic fields emanating from neodymium magnets can damage magnetic media such as floppy disks, credit cards, magnetic ID cards, cassette tapes, video tapes or other such devices. They can also damage older televisions, VCRs, computer monitors and CRT displays.What is neodymium used for?
Neodymium is used to make specialized goggles for glass blowers. The metal is also used in a spark producing alloy (misch metal) for cigarette lighter flints. Neodymium salts are used to color glasses and enamels.How long do neodymium magnets last?
Neodymium magnets are permanent magnets, and lose a fraction of their performance every 100 years if maintained within their optimum working conditions.How strong are neodymium magnets?
POWERFUL - Applied Magnets Rare Earth Neodymium Magnets have an estimated pull force of 70 lbs. and are triple layer coated (nickel-copper-nickel) for maximum durability and protection against corrosion.