How do you name Cycloalkanes with branches?
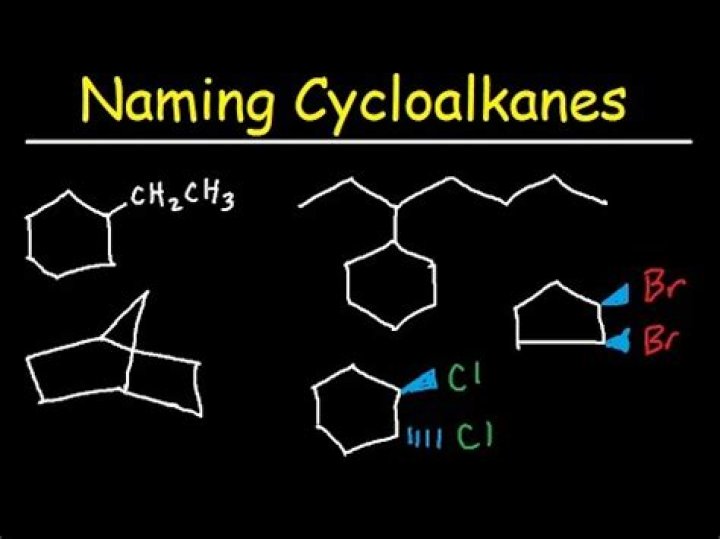
- Determine the cycloalkane to use as the parent chain. The parent chain is the one with the highest number of carbon atoms.
- If there is an alkyl straight chain that has a greater number of carbons than the cycloalkane, then the alkyl chain must be used as the primary parent chain.
.
Similarly one may ask, how do you name cyclic compounds?
A cyclic (ring) hydrocarbon is designated by the prefix cyclo- which appears directly in front of the base name. In summary, the name of the compound is written out with the substituents in alphabetical order followed by the base name (derived from the number of carbons in the parent chain).
Similarly, what is the formula of Cycloalkene? Those with two double bonds have the formula, CnH2n-2. Cycloalkenes have the general formula CnH2(n-m). The letter m represents the number of double bonds. Thus, cyclopropene has the formula C3H4 while that of cyclobutene is C4H6.
Subsequently, question is, what are Cycloalkanes give example?
Some of the common examples of cycloalkanes are the cyclopentane, Cyclobutane, cyclohexane, and cycloheptane, cyclooctane, etc as shown below in the image. The number of carbon atoms present in the compound decides the structure of cycloalkane.
How are Cycloalkanes formed?
The small cycloalkanes – in particular, cyclopropane – have a lower stability due to Baeyer strain and ring strain. These reactions are ring-opening reactions or ring-cleavage reactions of alkyl cycloalkanes. Cycloalkanes can be formed in a Diels–Alder reaction followed by a catalytic hydrogenation.
Related Question AnswersHow do you count Cycloalkanes?
Numbering the Cycloalkane When numbering the carbons of a cycloalkane, start with a substituted carbon so that thesubstituted carbons have the lowest numbers (sum). When two or more different substituents are present, number according to alphabetical order.What is a cyclic alkane?
Cyclic alkanes. In a cyclic alkane, we remove one of the hydrogens in each of the terminating methyl groups of a linear alkane chain and then use the dangling orbitals that result to form an additional bond between the ends to form a cyclic molecule.What is the name of the -- ch3 group?
A methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms — CH3. In formulas, the group is often abbreviated Me. Such hydrocarbon groups occur in many organic compounds. It is a very stable group in most molecules.What are the uses of Cycloalkanes?
Cycloalkanes can also be used for many different purposes. These uses are typically classified by the number of carbons in the cycloalkane ring. Many cycloalkanes are used in motor fuel, natural gas, petroleum gas, kerosene, diesel, and many other heavy oils.Are Cycloalkanes aromatic?
Explanation: Aromatic carbons have π bonds (Double Bonds) whereas Cycloalkanes don't have π bonds. Cycloalkanes on the other hand as depicted in the name "alkane" consist of a cyclic structure with n (n = 1,2,3,4) number of Carbon atoms but only have single bonds between the Carbons.Is benzene a Cycloalkene?
Benzene is an aromatic compound and Cycloalkene is an aliphatic cyclic compound. Yes benzene can be named as 1,3,5 cyclohexatriene . Aromatic compounds have different properties to that of alicyclic compounds hence we dont consider benzene as alicyclic system.What is a 5 carbon ring called?
The most common ring compounds contain either 5 or 6 carbons. These compounds are also called cyclic. Cyclopentane: Although the simplest representation is that of a line drawing of a pentagon as shown on the left.What is an example of nomenclature?
Use nomenclature in a sentence. noun. Nomenclature is defined as a system of names and terms used in a particular field of study or community. An example of nomenclature is the language of sculpture.Is alkane a functional group?
Alkanes are not usually considered as functional groups; instead, an alkane is a compound that lacks functional groups. The functional group in an alkene is a carbon-carbon double bond. The functional group in an alkyne is a carbon-carbon triple bond.Why is carbon important to life?
Most living things on Earth are made of carbon . Living things need carbon the most in order to live, grow, and reproduce. Also, carbon is a finite resource that cycles through the Earth in many forms. Also, carbon is so important to life because virtually all molecules in the body contain carbon.Why is Benzene not considered a Cycloalkene?
This is because Benzene varies significantly from alkenes in terms of chemical properties, so much so that it cannot be considered an alkene; this arises from the fact that due to the particular structure of Benzene, an important type of bonding occurs called delocalisation/conjugation occurs.What are the examples of alkanes?
For this reason, alkanes are not formally considered a functional group. In terms of occurrence and use, the simplest examples of alkanes are methane (CH4, the principal constituent of natural gas), ethane (C2H6), propane (C3H8, widely used as a gaseous fuel), and butane (C4H10, the liquid fuel in pocket lighters).What do u mean by nomenclature?
nomenclature. Nomenclature is a system for giving names to things within a particular profession or field. For instance, you may have heard of binomial nomenclature in biology class. It refers to the way of referring to living things by two names, like calling humans Homo sapiens.How are alkanes named?
The names of all alkanes end with -ane. Alkanes with unbranched carbon chains are simply named by the number of carbons in the chain. The first four members of the series (in terms of number of carbon atoms) are named as follows: CH4 = methane = one hydrogen-saturated carbon.What is the name of an 11 carbon chain?
List of straight-chain alkanes| Number of C atoms | Number of isomers | Name of straight chain |
|---|---|---|
| 9 | 35 | n-nonane |
| 10 | 75 | n-decane |
| 11 | 159 | n-undecane |
| 12 | 355 | n-dodecane |