So you se that strongly acidic [or strongly basic] solution will have high conductivity Since the pH is a measure of the concentration of the Hydrogen [and the Hydroxyl] ions, for an acidic solutin, the lower the pH [i.e. the higher the H+ concentration,] the greater the conductivity will be..
In this regard, are acids conductive?
Acids and bases in aqueous solutions will conduct electricity because they contain dissolved ions. Therefore, acids and bases are electrolytes. Strong acids and bases will be strong electrolytes. However, acids will react with metal, so testing conductivity may not be plausible.
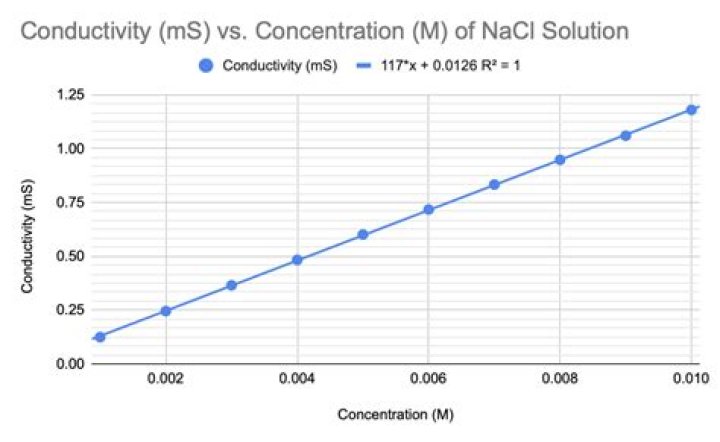
Likewise, what is the relationship between conductivity and concentration? Answer: Specific Conductivity decreases with a decrease in concentration. Since the number of ions per unit volume that carry current in a solution decrease on dilution. Hence, concentration and conductivity are directly proportional to each other.
Similarly, it is asked, what is acid conductivity?
Conductivity Principle: An acid or base which strongly conducts electricity contains a large number of ions and is called a strong acid or base. An acid or base which conducts electricity only weakly contains only a few ions and is called a weak acid or base.
What causes conductivity to increase?
Conductivity is a measure of the ability of water to pass an electrical current. Because dissolved salts and other inorganic chemicals conduct electrical current, conductivity increases as salinity increases. Conductivity is also affected by temperature: the warmer the water, the higher the conductivity.
Related Question Answers
Why is phenolphthalein pink?
Phenolphthalein (HIn) is weakly acidic in nature. And in aqueous solution, it dissociates into and ions. The pink colour of the solution is due to the concentration of ions in the solution. The pink colour of the solution is due to the concentration of ions in the solution.How do acids feel?
Acids taste sour while bases taste bitter. An acid reacts with metals to produce bubbles of hydrogen gas while a base feels slimy to the touch.Is acid good conductor of electricity?
Acids are good conductor of electricity when they are taken in aqueous solution. When an acid is dissolved in water,It lose hydrogen ions and loses lots of anions as well. Ionic solutions are good conductors of electricity.Does pH affect conductivity?
So you se that strongly acidic [or strongly basic] solution will have high conductivity Since the pH is a measure of the concentration of the Hydrogen [and the Hydroxyl] ions, for an acidic solutin, the lower the pH [i.e. the higher the H+ concentration,] the greater the conductivity will be.Is distilled water conductive?
Pure water is not a good conductor of electricity. Ordinary distilled water in equilibrium with carbon dioxide of the air has a conductivity of about 10 x 10-6 W-1*m-1 (20 dS/m). Because the electrical current is transported by the ions in solution, the conductivity increases as the concentration of ions increases.What are the strongest acids and bases?
11.8: Strong Acids and Bases
| Strong Acids | Strong Bases |
| HCl, HBr, HI HClO4 | O2– (Li2O, Na2O, K2O, Rb2O, Cs2O, CaO, SrO, and BaO are soluble |
| HNO3, H2SO4, HClO3 | H–, S2–, NH2–, N3–, P3– |
Are acids more reactive than bases?
Are acids more reactive than bases in general? From the standpoint of chemical reactivity there is absolutely no difference. Partially, it may stem form the property that concentrated acids are "hungry" for water and burn the skin by dehydration.Is Salt a good conductor of electricity?
This is because salt water is a good conductor of electricity. Salt molecules are made of sodium ions and chlorine ions. (An ion is an atom that has an electrical charge because it has either gained or lost an electron.) These ions are what carry electricity through water.Does pH affect voltage?
pH Electrodes At a pH of 7 (neutral), the electrodes will produce 0 volts between them. At a low pH (acid) a voltage will be developed of one polarity, and at a high pH (caustic) a voltage will be developed of the opposite polarity.Is HCl a strong acid?
A strong acid is an acid which is completely ionized in an aqueous solution. Hydrogen chloride (HCl) ionizes completely into hydrogen ions and chloride ions in water. A weak acid is an acid that ionizes only slightly in an aqueous solution. Because HCl is a strong acid, its conjugate base (Cl−) is extremely weak.Is NaOH an acid or base?
NaOH is a base because when dissolved in water it dissociates into Na+ and OH- ions. It is the OH- (hydroxyl ion) which makes NaOH a base. In classical term a base is defined as a compound which reacts with an acid to form salt and water as depicted by the following equation. NaOH+HCl=NaCl+H2O.Is vinegar acidic or basic?
Summary Vinegar is mildly acidic with a pH of 2–3. Apple cider vinegar is slightly more alkaline than pure vinegar because it contains more alkaline nutrients. However, it's still acidic.Is hno3 a strong acid?
The 7 common strong acids are: HCl, HBr, HI, HNO3, HClO3, HClO4 and H2SO4 (1st proton only). They simply do not ionize completely whereas a strong acid or base does. That means for a solution of 0.1 M HCl compared to 0.1 M HF the HCl solution will contain more H+ and be more acidic.What makes a strong acid?
A strong acid is any acid that ionizes completely in solutions. This means it gives off the greatest number of hydrogen ions or protons when placed in a solution. Ions are charged particles. This means that it has one proton, and therefore a hydrogen ion is essentially a proton.What is pH and conductivity?
pH is the negative logarithm of Hydrogen ion concentration. Conductivity is a measure of the ability of a material to conduct electricity. The two concepts are not generally related. For instance, a pure copper wire has a high conductivity while containing no Hydrogen ions.Is NaOH a strong base?
Sodium hydroxide (NaOH) is strong base because it fully dissociates in water to produce hydroxide ions. While ammonia (NH3) is weak base because it accepts protons from water to produce fewer hydroxide ions in solution. While weak bases produce fewer hydroxide ions, making the solution less basic.Is lemon juice an acid or base?
Lemon juice in its natural state is acidic with a pH of about 2, but once metabolized it actually becomes alkaline with a pH well above 7. So, outside the body, anyone can see that lemon juice is very acidic. However, once fully digested, its effect is proven to be alkalizing with many health benefits.What does conductivity depend on?
Factors that affect the conductivity of electrolytes (ESAFQ) The conductivity of an electrolyte is therefore affected by the following factors: The concentration of ions in solution. The higher the concentration of ions in solution, the higher its conductivity will be. The type of substance that dissolves in water.Does salt increase conductivity?
In general, the conductivity of salt solutions increases as the amount of dissolved salt increases. The exact increase in conductivity, however, is complicated by the relationship between the concentration of the salt and the mobility of its charged particles.