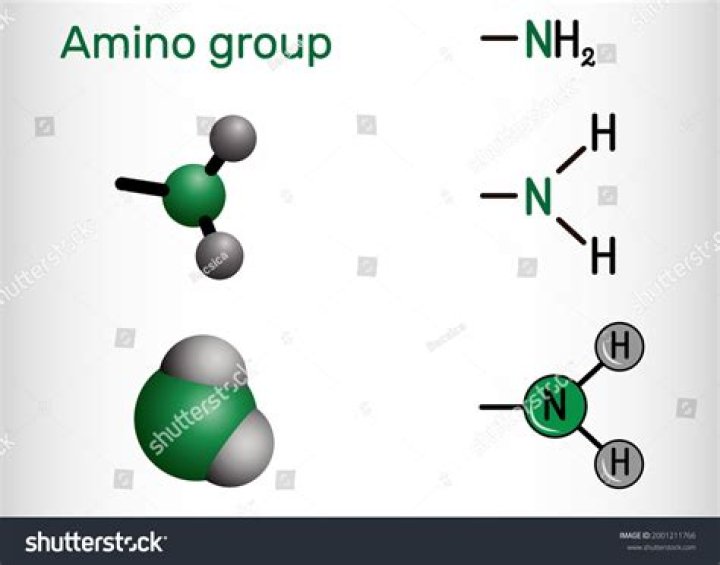
amino group
.
Besides, what is a nh2 group called?
Yes, -NH2 is a functional group! This group is also called as the amine or the amino group. This group is obtained by replacing one atom hydrogen of the ammonia molecule by an aryl group or an alkyl.
Also Know, how many types of amine are there? Amines are classified as primary, secondary, or tertiary depending on whether one, two, or three of the hydrogen atoms of ammonia have been replaced by organic groups. In chemical notation these three classes are represented as RNH2, R2NH, and R3N, respectively.
Similarly, it is asked, what is the difference between a primary secondary and tertiary amine?
The main difference between primary secondary and tertiary amines is that, in primary amines, one alkyl or aryl group is attached to the nitrogen atom and in secondary amines, two alkyl or aryl groups are attached to the nitrogen atom whereas, in tertiary amines, three alkyl or aryl groups are attached to the nitrogen
How do you identify amine groups?
Amines are classified according to the number of carbon atoms bonded directly to the nitrogen atom. A primary (1°) amine has one alkyl (or aryl) group on the nitrogen atom, a secondary (2°) amine has two, and a tertiary (3°) amine has three (Figure 15.10.
Related Question Answers
What functional group is COOH?
A carboxyl group (COOH) is a functional group consisting of a carbonyl group (C=O) with a hydroxyl group (O-H) attached to the same carbon atom.What is amine formula?
The basic chemical structure is that of ammonia (NH3) with the key atom being the central nitrogen atom. Try to remember that an amine is just like ammonia because ammonia is a simple molecule to recall. Primary amines are called things like methylamine (CH3-NH2) and ethylamine (CH3-CH2-NH2).What are aryl groups?
An aryl group is a functional group derived from a simple aromatic ring compound where one hydrogen atom is removed from the ring. Usually, the aromatic ring is a hydrocarbon. Examples: The phenyl functional group (C6H5) is an aryl functional group derived from benzene.Is benzene a functional group?
Benzene ring: An aromatic functional group characterized by a ring of six carbon atoms, bonded by alternating single and double bonds. A benzene ring with a single substituent is called a phenyl group (Ph). Benzene has a benzene ring, but pyridine does not.Are amines chiral?
Amines of the type NHRR′ and NRR′R″ are chiral: the nitrogen center bears four substituents counting the lone pair. Because of the low barrier to inversion, amines of the type NHRR′ cannot be obtained in optical purity. Inversion of an amine.Who discovered Amine?
Casimir
Is h3n a functional group?
First and foremost, NH3 is not a functional group. It's a compound called Ammonia. However, the variant where it functions as a functional group is when either one, two or even three of the Hydrogen accompanying the Nitrogen atom is replaced by a carbon moeity. It's a compound called Ammonia.Is a hydroxyl group?
Hydroxyl groups are a functional group found in sugars and alcohols. A hydroxyl group consists of one hydrogen and one oxygen atom and can be written as either -OH or HO-. Hydroxyl groups are polar, and the oxygen side is always negative, while the hydrogen side is always positive.What is amine used for?
The most commonly used amines in industrial plants are the alkanolamines DEA, MEA, and MDEA. These amines are also used in many oil refineries to remove sour gases from liquid hydrocarbons such as liquified petroleum gas (LPG).How do you classify amines primary secondary and tertiary?
Amines are classified as primary, secondary, or tertiary according to the number of carbons bonded directly to the nitrogen atom. Primary amines have one carbon bonded to the nitrogen. Secondary amines have two carbons bonded to the nitrogen, and tertiary amines have three carbons bonded to the nitrogen.What does an amine do?
3.9 Amines A quaternary ammonium ion; the R designations represent alkyl groups or aryl groups. Amines are particularly valuable because of their ability to act as bases, a property that is a consequence of the ability of amines to accept hydrogen atoms from acidic molecules.Which amine is most basic?
The inductive effect makes the electron density on the alkylamine's nitrogen greater than the nitrogen of ammonium. Correspondingly, primary, secondary, and tertiary alkyl amines are more basic than ammonia.What is a primary amine?
A primary (1º) amine is an amine that has the following general structural formula. R= alkyl, aryl. eg: The NH2 group in a primary amine molecule is called the primary amine group. See also secondary amine, tertiary amine.How do you name a tertiary amine?
Primary amines are attached to one alkyl, or carbon group, secondary amines are attached to two alkyl groups, and tertiary amines are attached to three alkyl groups. To name amines, use the IUPAC name for the longest carbon chain attached and exchange the 'e' for the word amine.Is ethylamine a primary amine?
In primary amines, only one of the hydrogen atoms in the ammonia molecule has been replaced. For example, the simplest amine, CH3NH2, can be called methylamine, methanamine or aminomethane. The commonest name at this level is methylamine and, similarly, the second compound drawn above is usually called ethylamine.What is an amide group?
An amide is a functional group containing a carbonyl group linked to a nitrogen atom or any compound containing the amide functional group. Amides are derived from carboxylic acid and an amine.Which foods are high in amines?
Vasoactive amines such as tyramine, serotonin and histamine are well known triggers of migraines in some people. They are present naturally in pineapples, bananas, baked meat, vegetables, red wine, wood-matured white wine, avocados, chocolate, citrus fruits and mature cheese.Are primary amines soluble in water?
Primary amines with three or four carbon atoms are liquids at room temperature whereas higher ones are solids. Aniline and other arylamines are generally colourless. Lower aliphatic amines can form hydrogen bonds with water molecules. Therefore, such amines are soluble in water.Are amines weak bases?
Amines, unless they have four R- groups attached (a quaternary amine) have a lone pair of electrons, which just like the lone pair in ammonia, can accept a proton. Hence amines, like ammonia, are weak bases. Carboxylic acids biochemistry's weak acids.