Why is sf6 an octahedral?
.
Herein, why is sf6 nonpolar?
SF6 has an octahedral molecular geometry, which means that the sulfur molecule has six fluorine atoms surrounding it. While each individual bond is polar, there is no net effect, meaning that the molecule is nonpolar. Because there are six fluorine atoms, this means that each atom is 90 degrees from its neighbors.
One may also ask, what is the molecular geometry for sf6? The molecular geometry of SF6 is octahedral with a symmetric charge distribution around the central atom.
Hereof, is sf6 covalent?
Because fluorine forms strong covalent bonds, the S-F bonds have high bond energies and are difficult to break. Because there are so many fluorine atoms surrounding the sulphur atom, it is well shielded from attacking molecules too.
What is the hybridization of sf6?
The S atom in SF6 undergoes sp3d2 hybridization which involves one s orbital, three p orbitals,and two d orbitals. The resulting hybrid is as follows.
Related Question AnswersIs h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.How many lone pairs are in sf6?
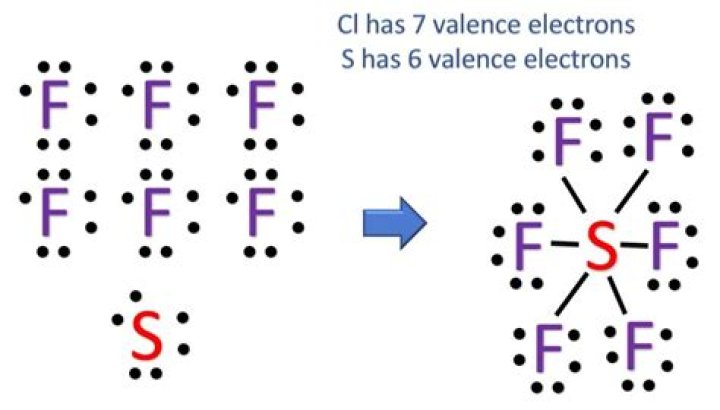
SF6 molecular geometry. Sulfur hexafluoride has a central sulfur atom around which one can see 12 electrons or 6 electron pairs. Thus, the SF6 electron geometry is considered to be octahedral. All the F-S-F bonds are 90 degrees, and it has no lone pairs.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.Is BCl3 polar or nonpolar?
The molecular geometry of BCl3 is trigonal planar with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.Does so2 have a dipole moment?
The SO2 molecule has a dipole moment. This molecule does not have a permanent dipole moment (i.e., the dipole moment of CO2 is zero). The requires the molecule to be linear because oxygen is more electronegative than carbon and therefore the C=O. bond will be polarized.Is CCl4 polar or nonpolar?
CCl4 is an example of a nonpolar molecule. The four bonds of carbon tetrachloride (CCl4) are polar, but the molecule isnonpolar because the bond polarity is canceled by the symmetric tetrahedral shape. When other atoms substitute for some of the Cl atoms, the symmetry is broken and the molecule becomes polar.What type of bond is sulfur hexafluoride?
The structure has a Sulfur atom in the middle with 6 Fluorine atoms surrounding this structure. Each fluorine atom has 3 unshared pairs to achieve the octet rule. In addition, this bond is polar covalent.What type of bond is mgf2?
Magnesium Fluoride is an ionic compound. The magnesium atom gives up 2 electrons to form a magnesium ion, Mg2+ . The two electrons are transferred to fluorine atoms to form two fluoride ions, F–. The magnesium fluoride has the formula MgF2.How many bonding electrons does sf6 have?
12 bonding electronsIs h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.Is c2h4 polar or nonpolar?
In C2H4, due to small electronegativity difference between C and H , the C-H bond is slightly polar but the molecule is symmetrical around its centre line which results in even charge distribution and makes the molecule non- polar.What is the bond angle of bf3?
In BF3, the boron atom has no lone pair, so the molecule adopts the ideal trigonal planar geometry with ideal 120 degree bond angles.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.



