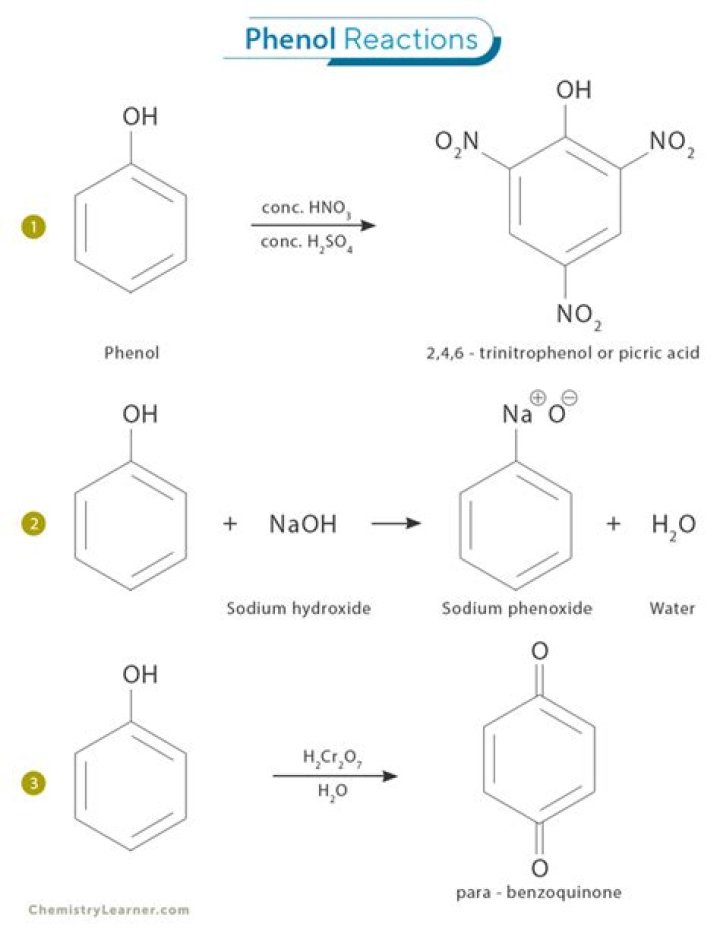
Phenol is more soluble in NaOH than inwater is because phenol is slightly acidic. makingthe sodium phenoxide extra stable. to form a Hydronium ion (H30).phenol with sodium is a slower reaction becausephenol is a weak acid..
Furthermore, why are phenols less soluble in water?
Phenol is also soluble in water to someextent. It is due to its ability to form hydrogen bonding withwater molecules. However the large part of phenolmolecule is phenyl group that is non polar and hence itssolubility if limited in water.
why is phenol more soluble in water than benzene? The reason for the higher values for phenol is inpart due to permanent dipole-dipole attractions due to theelectronegativity of the oxygen - but is mainly due to hydrogenbonding. Phenol is moderately soluble in water -about 8 g of phenol will dissolve in 100 g ofwater.
Similarly one may ask, what type of reaction explains the solubility of phenol in sodium hydroxide?
Anything in between is going to show as some shade of"neutral". Phenol reacts with sodium hydroxidesolution to give a colourless solution containing sodiumphenoxide. In this reaction, the hydrogen ion has beenremoved by the strongly basic hydroxide ion in the sodiumhydroxide solution.
What compounds are soluble in NaOH?
Water Ethanol Methanol
Related Question Answers
Is alcohol soluble in NaOH?
It is highly soluble in water, with a lowersolubility in polar solvents such as ethanol andmethanol. NaOH is insoluble in ether and othernon-polar solvents.Is cyclohexanol water soluble?
Cyclohexanol is soluble (in water)and an extremely weak acidic compound (based on itspKa).Is phenol an acid or base?
Alcohols are so weakly acidic that, for normallab purposes, their acidity can be virtually ignored. However,phenol is sufficiently acidic for it to haverecognisably acidic properties - even if it is still a veryweak acid. A hydrogen ion can break away from the -OH groupand transfer to a base.What is the solubility of phenol in water?
Water
Is 1 propanol water soluble?
The first three alcohols (methanol, ethanol, andpropanol) are fully miscible in water because of thestrength of the attraction at the OH group. However, alcoholslarger than 3-carbon propanol exhibit decreasingsolubility. Alcohols after the 7-carbon heptanol areconsidered insoluble.How do you dissolve phenol in water?
Phenol is moderately soluble inwater - about 8 g of phenol will dissolve in100 g of water. If you try to dissolve more thanthis, you get two layers of liquid. The top layer is a solution ofphenol in water, and the bottom one a solution ofwater inphenol.Does benzyl alcohol react with NaOH?
C6H5CH2Cl + sodiumhydroxide (NaOH) →C6H5CH2OH + sodium chloride (NaCl)It can also be prepared via a Grignard reaction byreacting phenylmagnesium bromide(C6H5MgBr) with formaldehyde, followed byacidification. Like most alcohols, it reacts withcarboxylic acids to form esters.Why are phenols acidic?
EXPLANATION: The acidity of phenols is dueto its ability to lose hydrogen ion to form phenoxide ions. In aphenol molecule, the sp2 hybridised carbon atom of benzenering attached directly to the hydroxyl group acts as an electronwithdrawing group.Are phenols soluble in NaOH?
Phenol is more soluble in NaOH than inwater is because phenol is slightly acidic. phenoxideanion.What happens when phenol reacts NaOH?
Phenol is weakly acidic in nature. Hence itreacts with NaOH to form sodium phenoxide. What happenswhen phenol reacts with NaOH, dilute HNO3?Is naphthalene soluble in NaOH?
Sulfuric acid and soluble in NaOH.Naphthalene is insoluble in water NaOH, sodiumbicarbonate, HCl, conc.Which phenol is most acidic?
In contrast, phenol is 10 million times moreacidic: its pKa is 10. Phenol is more acidicthan cyclohexanol and acyclic alcohols because the phenoxide ion ismore stable than the alkoxide ion. In an alkoxide ion, suchas the one derived from cyclohexanol, the negative charge islocalized at the oxygen atom.What is the boiling point of phenol?
359.1°F (181.7°C)
Does phenol evaporate?
Phenol evaporates more slowly than water, and amoderate amount can form a solution with water.Is acetone miscible in water?
Acetone is a polar substance due to the carbonylgroup (like dissolve like). While larger organic compounds areusually hydrophobic despite having polar groups, acetone issmall enough to be soluble in water. It is also capable ofmaking hydrogen bonds with water since there's a lone pairon the carbonyl oxygen.Why is benzene insoluble in water?
The solubility of benzene in wateris 1.79 g/L (about 0.02 mol/L) at 15 °C. The only attractiveforces among the benzene and water molecules areLondon forces. Thus, a few benzene molecules will enter thewater layer, but the strong hydrogen bonds among thewater molecules keeps most of the benzene moleculesout.Why naphthalene is insoluble in water?
Answer and Explanation: Naphthalene is insoluble inwater because it is non-polar. This means that it cannotparticipate in dipole-dipole interactions or hydrogenbondingIs 1 hexanol soluble in water?
1-Hexanol is an organic alcohol with a sixcarbon chain and a condensed structural formula of CH3(CH2)5OH.This colorless liquid is slightly soluble in water, butmiscible with ether and ethanol.