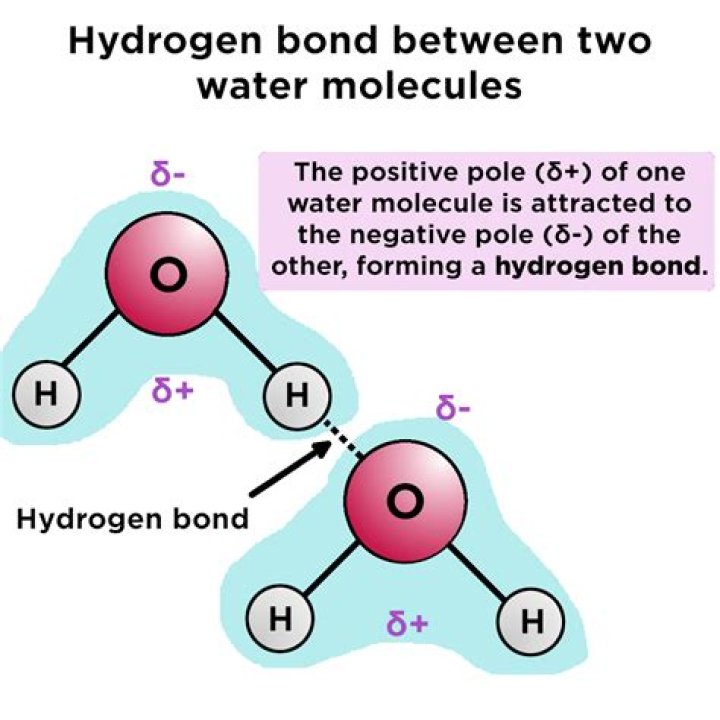
Hydrogen bonds in water provide many characteristic benefits to water: cohesion (holding water molecules together), high specific heat (absorbing heat when breaking, releasing heat when forming; minimizing temperature change), high heat of vaporization (several hydrogen bonds must be broken in order to evaporate water).
Similarly, you may ask, how do hydrogen bonds affect water's properties?
Water's high heat capacity is a property caused by hydrogen bonding among water molecules. When heat is absorbed, hydrogen bonds are broken and water molecules can move freely. When the temperature of water decreases, the hydrogen bonds are formed and release a considerable amount of energy.
Furthermore, what would happen without hydrogen bonding? Hydrogen bonds ensures the reduction of temperature extremes in large bodies of water. However, without hydrogen bonds, water in oceans and lakes would begin to rapidly boil due to the vast decrease in boiling point – resulting in massive problems for life on Earth.
Also know, why is hydrogen bonding in water important to life?
The presence of hydrogen bonds also makes water molecules more 'sticky' or in scientific terms cohesive and adhesive. The small charges on the water molecules allows them to stick together which is why water has a 'skin' that small insects can walk on, and also explains why water can be sucked up a straw so easily.
What are the 3 most important properties of water?
The main properties of water are its polarity, cohesion, adhesion, surface tension, high specific heat, and evaporative cooling. A water molecule is slightly charged on both ends. This is because oxygen is more electronegative than hydrogen.
Related Question Answers
What type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.Why are hydrogen bonds so important?
Hydrogen bonding is important in many chemical processes. Hydrogen bonding is responsible for water's unique solvent capabilities. Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.How are hydrogen bonds formed?
A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. Hydrogen has one proton and one electron. This makes hydrogen an electrically positive atom because it has a deficiency of electrons. What are the properties of hydrogen bonding?
Properties of hydrogen bonds. a hydrogen bond is formed when a charged part of a molecule having polar covalent bonds forms an electrostatic (charge, as in positive attracted to negative) interaction with a substance of opposite charge. Molecules that have nonpolar covalent bonds donot form hydrogen bonds.What is the most important role of hydrogen bonding between water molecules?
Hydrogen bonds in water provide many characteristic benefits to water: cohesion (holding water molecules together), high specific heat (absorbing heat when breaking, releasing heat when forming; minimizing temperature change), high heat of vaporization (several hydrogen bonds must be broken in order to evaporate water)Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.Which bond is the strongest?
ionic bond
What do hydrogen bonds do?
Hydrogen Bonds. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. The attraction created by hydrogen bonds keeps water liquid over a wider range of temperature than is found for any other molecule its size.What properties make water important to life?
Water is essential for all living things. Water's unique density, high specific heat, cohesion, adhesion, and solvent abilities allow it to support life.Why is it important that hydrogen bonds are weak?
Weak bonds may be easily broken but they are very important because they help to determine and stabilize the shapes of biological molecules. Hydrogen bonds keep complementary strands of DNA together. Hydrogen bonds participate in enzymic catalysis.Is water a hydrogen bond?
Hydrogen-bonding forms in liquid water as the hydrogen atoms of one water molecule are attracted towards the oxygen atom of a neighboring water molecule; generally, a proton shared by two lone electron pairs. This attraction is the basis of the 'hydrogen' bonds.Is water a covalent bond?
H2O or water as it is more commonly known as is a molecule consisting of 2 Hydrogen molecules bonded to one Oxygen molecule. As the table shows this makes H2O a molecule with a polar covalent bond. Well, electronegativity is the measure of how attracted bond seeking electrons are to an element.Where are hydrogen bonds found?
A ubiquitous example of a hydrogen bond is found between water molecules. In a discrete water molecule, there are two hydrogen atoms and one oxygen atom.Is hydrogen essential to life?
The hydrogen atom has one proton and one electron. The most abundant element in the universe, hydrogen is also a promising source of "clean" fuel on Earth. Hydrogen is essential for life, and it is present in nearly all the molecules in living things, according to the Royal Society of Chemistry.What type of bond is a hydrogen bond?
A hydrogen bond is the electromagnetic attraction between polar molecules in which hydrogen is bound to a larger atom, such as oxygen or nitrogen. This is not a sharing of electrons, as in a covalent bond. Instead, this is an attraction between the positive and negative poles of charged atoms.Are hydrogen bonds strong?
Hydrogen bonds are strong intermolecular forces created when a hydrogen atom bonded to an electronegative atom approaches a nearby electronegative atom. The hydrogen bond is one of the strongest intermolecular attractions, but weaker than a covalent or an ionic bond.What is hydrogen bonding and its types?
Hydrogen bond or hydrogen bonding is a type of weak force that results in the formation of dipole-dipole interaction between a hydrogen atom and an electronegative atom that is strongly bonded to another electronegative atom. However, they are weak compared to true covalent or ionic bonds.Is ch4 a hydrogen bond?
Answer and Explanation: CH4 cannot form hydrogen bonds. This is because hydrogen bonds are a type of electrostatic interaction, which is only possible in molecules in whichWhy is polarity important to life?
Water's polarity allows it to dissolve other polar substances very easily. Wherever water goes, it carries dissolved chemicals, minerals, and nutrients that are used to support living things. Because of their polarity, water molecules are strongly attracted to one another, which gives water a high surface tension.