why H2SO4 has an exceptional lewis structure? Dear Student, H2SO4 has an exceptional Lewis diagram because here the the number of electrons shared by S becomes 12 i.e. its exceeds the octet rule. Some have electrons less than octet and some has more than octet..
Considering this, what is the Lewis dot structure of h2so4?
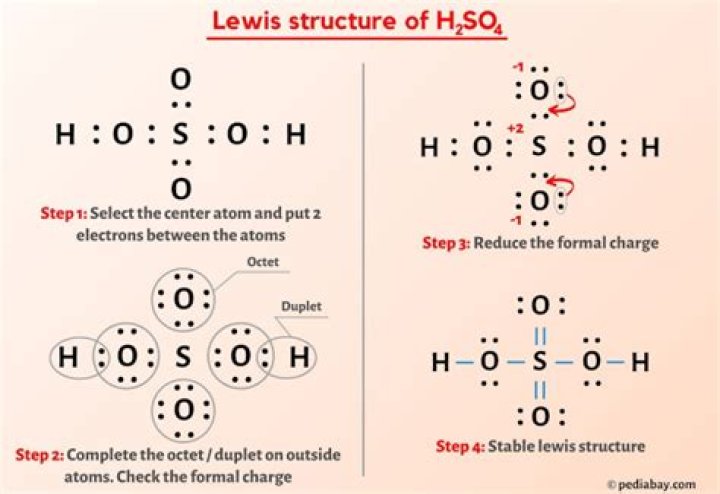
a step -by step explanation of how to draw the h2so4 lewis structure (sulfuric acid.) when we have an h(or h2)in front of a polyatomic molecule( like co3,so4,no2,etc.) we know that it's an acid. this means that the hydrogen atoms will be attached to the outside of the oxygen molecules.
One may also ask, what type of bond is present in h2so4? If you want the "Intro to Chemistry" answer, then H2SO4 has a distinct ionic bond between H+ and HSO4-. It is hygroscopic (absorbs water from the air), so it almost always exists as a solution in water (at least in practical use). Sulfuric acid is a strong acid, so the two ions are dissociated in water.
In this way, is h2so4 polar or nonpolar?
Sulfuric acid (H2SO4) is a strong mineral. Anhydrous sulfuric acid is a very polar liquid.
Why are Lewis dot structures important?
Lewis structures (also known as Lewis dot structures or electron dot structures) are diagrams that represent the valence electrons of atoms within a molecule. These Lewis symbols and Lewis structures help visualize the valence electrons of atoms and molecules, whether they exist as lone pairs or within bonds.
Related Question Answers
What is the structure of HClO4?
Where n in this case is 5 since HClO4 consists of 6 atoms but one of them is a hydro gen atom (remember n is the number of atoms in a molecule minus the hydrogen atoms). So there are no π electrons in HClO4 and therefore the structure of Step 1 is the Lewis structure.Which is the Lewis structure for h3po4?
There are a total of 32 valence electrons for the H3PO4 Lewis structure. When we have an H (or H2 or H3) in front of a polyatomic molecule (like CO3, SO4, NO2, PO4,etc.) we know that it's an acid.What is the structure of h2s2o8?
Peroxydisulfuric acid is the inorganic compound with the chemical formula H2S2O8. Also called Marshall's acid after its inventor Professor Hugh Marshall, it is a sulfur oxoacid. In structural terms it can be written HO3SOOSO3H. It contains sulfur in its +6 oxidation state and a peroxide group.What is the structure of h2s2o7?
H2S2O7 crystallizes in the space group C2/c with a = 1295.5(8) pm, 6 = 1370.5(7) pm, c = 1316.4(8) pm, ß = 109.20(5)°, and Z = 16 formula units. The structure is characterized by slightly eclipsed H2S2O7 molecules being linked by hydrogen bonds.What is the formula for h2so4?
Originally Answered: What is the sulfuric acid formula? Sulphuric Acid's Chemical Formula is H2SO4. Or, to be more clear: 2 Hydrogen Atoms and 1 Sulfate Molecule (1 Sulfur Atom and 4 Oxygen Atoms). It has a Molecular Weight of 98.072 g/mol.What is s2ho4?
Chemical name of (H2SO4) is sulfuric acid also called oil of vitriol, or hydrogen sulphate. It is a very strong acid; Sulfuric acid is a colorless oily liquid. It is soluble in water with release of heat.What is the Lewis structure for CO?
Drawing the Lewis Structure for CO The Lewis structure for CO has 10 valence electrons. For the CO Lewis structure you'll need a triple bond between the Carbon and Oxygen atoms in order to satisfy the octets of each atom while still using the 10 valence electrons available for the CO molecule.What is the Lewis structure for hno3?
The HNO3 Lewis structure is best thought of as the NO3 with an H attached to one of the oxygen atoms. This is a pattern seen with many acids. For the HNO3 Lewis structure, calculate the total number of valence electrons for the HNO3 molecule.Is HCL polar or nonpolar?
There actually are simple HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. HCL is neither polar or non-polar.Is h2so4 aqueous?
Sulfuric acid is a very strong acid; in aqueous solutions it ionizes completely to form hydronium ions (H3O+) and hydrogen sulfate ions (HSO4−).Is benzene polar or nonpolar?
We could say that benzene is nonpolar because it is a hydrocarbon that contains only C-C and C-H bonds, and hydrocarbons are nonpolar. But C is slightly more electronegative than H (by 0.35 units), so a C-H bond is very slightly polar and has a small dipole moment.Is h2so4 an acid or base?
Absolutely H2SO4 is acid. It's acidic nature is due to H+ ion present in them. When a molecule of H2SO4 is comes in the contact of water it breaks down into H+ and HSo4- ion. At first we have to understand that we say it is an acid or base relative to another base and acid , a substance is an acid w.r.t a base .How many atoms are in h2so4?
One sulfuric (H2SO4) molecule has 2 hydrogen atoms, 1 sulfur atom, and 4 oxygen atoms. You can also say one mole of sulfuric acid has two mols of hydrogen atoms, 1 mol of sulfur atoms, and 4 moles of oxygen atoms. So in total, we have 7 mols of atoms. But that's in 1 mol of sulfuric acid.What type of bond is NaCl?
Salt or Sodium Chloride (NaCl) is a good example of a ionic bonding. Sodium (Na) has 1 valance electron and Chlorine (Cl) has 7 electrons in its outer orbit. If Sodium lost its valance electron, its next shell will be full. But that would also make Sodium a positive ion.Is sulfuric acid a covalent or ionic bond?
If you look at all the bonds in H2SO4 in terms of electronegativity difference, all of them are covalent (polar). The O-H bond in sulfuric acid is very weak. That does not make it an ionic bond. Just because it readily forms ions in water does not make it an ionic bond.Is h2so3 an acid or base?
The hydrogen sulfite, or bisulfite, ion is the ion HSO3-. It is the conjugate base of sulfurous acid, H2SO3.Is HCl ionic or molecular?
But, when HCl is dissolved in water, it readily dissociated into H+ and Cl- ions. These ions can move through the solution and that makes HCl an ionic compound. So, this molecule can be classified as either covalent or ionic depending on the state of the molecule.Is HCl ionic or covalent?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.Is h3po4 an acid or base?
Salts: AgNO3(Salt), NaCl(Salt), NaNO3(salt), KNO3(salt), Acids: H3PO4(acid), HCl(acid), H2SO4(acid), HC2H3O2(acid), HNO3(acid), H2SO3(acid) Bases: Mg(OH)2(base), NaOH(base), KOHbase), NH4OH(base).