Why Electrovalent compounds are soluble in water?
.
Hereof, why Electrovalent compounds are insoluble in kerosene?
Water is a polar covalent compound. It decreases the electrostatic forces of attraction, resulting in free ions in aqueous solution. Hence, ionic compounds dissolve in water. Kerosene is a non-polar organic solvent and dissolves non-polar covalent compounds.
Likewise, why NaCl is soluble in water but not in kerosene? As sodium chloride is polar molecule it will be soluble in polar solvents like water. And insoluble in kerosene (as it is non-polar solvent). salt dissolve in water but not in kerosene because Kerosene comes from petroleum which is a nonpolar hydrocarbon that does not mix with water.
Simply so, why ionic compounds are soluble in water?
Most ionic compounds are soluble in water. This is because polar water molecules have a strong attraction for charged ions. The charged ions become solvated as they dissociate into water.
Why do Electrovalent compounds exist as solid?
Crystal Structure: In the solid state of electrovalent compounds, anions and cations are arranged in a regular manner. Melting Point and Boiling Point: Electrovalent or ionic compounds have high melting and boiling points because they need a large amount of energy to break strong ionic bonds.
Related Question AnswersWhat can dissolve in kerosene?
Kerosene is a hydrocarbon obtained from the fractional distillation of coal tar at temperature about 150 and 275 °C. Since it is a hydrocarbon, it is considered as a non-polar solvent. Oils and fats are common examples of non-polar compounds. Therefore these are very good soluble in kerosene.Is ionic bond soluble in water?
Ionic compounds dissolve in water if the energy given off when the ions interact with water molecules compensates for the energy needed to break the ionic bonds in the solid and the energy required to separate the water molecules so that the ions can be inserted into solution.Which things are soluble in petrol?
Petroleum solvents readily dissolve all petroleum fractions, vegetable oils and fats, and organic compounds of sulfur, oxygen, and nitrogen.Why covalent compounds are insoluble in water?
When covalent compounds dissolve in water they break apart into molecules, but not individual atoms. Water is a polar solvent, but covalent compounds are usually nonpolar. This means covalent compounds typically don't dissolve in water, instead making a separate layer on the water's surface.Are ionic compounds good conductors of electricity?
Ionic compounds are solids with high melting and boiling points. They are good conductors of electricity but only when dissolved in water.What is non polar solvent?
Polar solvents have large dipole moments (aka “partial charges”); they contain bonds between atoms with very different electronegativities, such as oxygen and hydrogen. Non polar solvents contain bonds between atoms with similar electronegativities, such as carbon and hydrogen (think hydrocarbons, such as gasoline).What is the meaning of polar solvent?
A polar solvent is a liquid with molecules that have a slight electrical charge due to its shape. For example, water is a molecule with one oxygen and two hydrogen atoms. The two hydrogen atoms are not on opposite sides of the oxygen, but rather at an angle.Are ionic compounds soluble in organic solvents?
Ionic compounds are soluble in water but not in organic solvent since water is a polar compound which decreases the electrostatic forces if attraction, resulting in free ions in aqueous solution. Hence they dissolve.Are all ionic compounds are soluble in water?
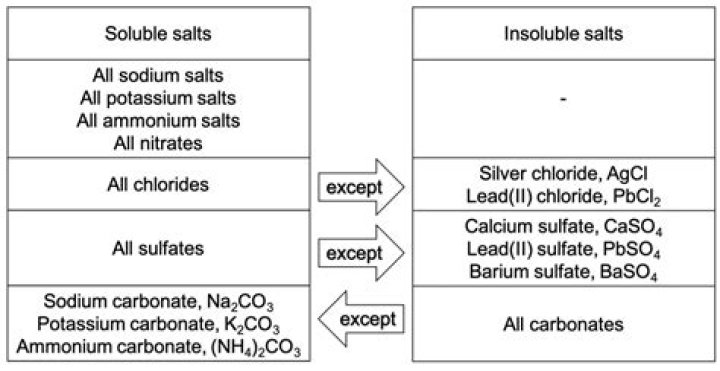
All ionic compounds are soluble in water to some extent, but the degree of solubility varies. While some compounds dissolve almost completely, others dissolve to such a small extent that they are simply called insoluble compounds. Such compounds include calcium sulfate, silver chloride, and lead hydroxide.Why are ionic bonds so weak in water?
Ionic bonds within a salt crystal may be quite strong. However, if the crystal of salt is dissolved in water, each of the individual ions becomes surrounded by water molecules, which inhibit oppositely charged ions from approaching one another closely enough to form ionic bonds. In water, ionic bonds are very weak.Is water an ion?
Likewise, a water molecule is ionic in nature, but the bond is called covalent, with two hydrogen atoms both situating themselves with their positive charge on one side of the oxygen atom, which has a negative charge.What is the solubility of ionic compounds?
Ionic compounds are most soluble in polar solvents like water, because the ions of the solid are strongly attracted to the polar solvent molecules. Ionic compounds are less soluble is solvents that contain a common ion. For example, CaSO4 is slightly soluble in water.What ions are soluble in water?
| Soluble Compounds | Exceptions |
|---|---|
| All salts of Cl-, Br- and I- | Halides of Ag+, Hg2 + and Pb2+ |
| Compounds containing F- | Flourides of Mg 2+, Ca 2+, Sr 2+, Ba 2+, Pb 2+ |
| Salts of nitrate, NO -3, Chlorate, ClO3 - perchlorate, ClO4 - acetate, CH3CO2 - | |
| Slats of sulfate, SO4 2- | Sulfates of Sr 2+, Ba 2+, Pb 2+ |



