Hot air balloons rely on the fact that the density of air decreases as it gets hotter. A gas burner, mounted above a balloon's basket, is used to heat the air inside the balloon. As the air heats, it increases in volume, or expands. The hot air balloon rises because its average density is less than the surrounding air..
Also, what happens to the density of gas as it is heated?
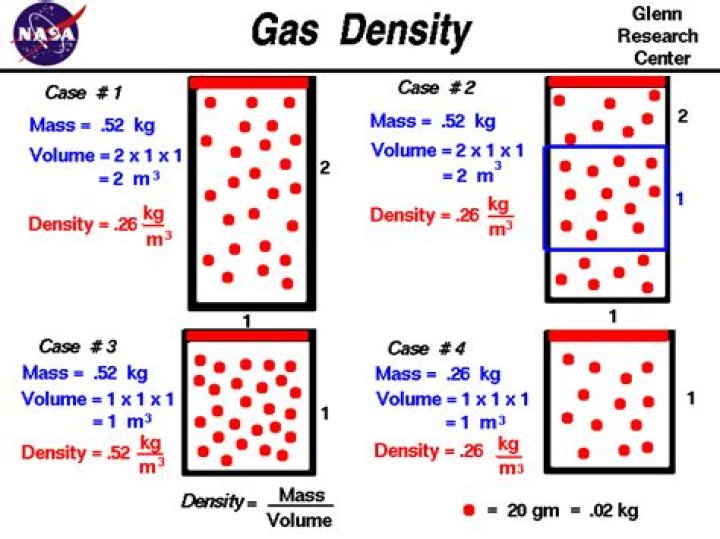
simple example is hot air balloon works on the same principle. This is because as temperature increases gas expands thus it's volume increases for same mass and density of the gas decreases as a result. Thus, density of the gas decreases as temperature increases.
Beside above, when a gas is heated at constant pressure its density? So, as volume in this process depends upon temperature so as we increase temperature volume also increases. Now as density is inversely proportional to volume so for the given mass of gas density decreases as volume increases. Thus for a given mass if gas is heated at constant pressure then it's density decreases.
Herein, what happens to the density of air in a balloon when it is heated?
As air is heated, it becomes less dense than the surrounding cooler air. The less dense hot air has enough lifting power to cause the balloon to float and rise into the air.
How does Charles law affect hot air balloons?
Charles's Law says that the volume of a gas is directly related to the temperature of that gas, similarly when a gas is heated, like a burner in a hot air balloon, the gas expands. So when the air inside the balloon expands, it becomes less dense and provides the lift for the hot air balloon.
Related Question Answers
Does temperature affect density of gas?
When a liquid or gas is heated, the molecules move faster, bump into each other, and spread apart. Because the molecules are spread apart, they take up more space. They are less dense. Therefore temperature can affect density.What happens when you remove heat from a gas?
If heat is removed from water vapour, the gas cools down and it condenses back into liquid water. Continue to cool the water (by removing heat), and it becomes solid ice. This is its freezing point.What happens when gas is cooled?
What Happens to a Gas When It Is Cooled? The volume, or the space that a particular gas occupies, decreases when it is cooled. The ideal gas equation indicates that decreasing the temperature causes a reduction in its volume. When a gas is cooled down, the atoms comprising the gas have less energy to move around.How does heat affect gas?
When you heat a gas, both its vapor pressure and the volume it occupies increase. The individual gas particles become more energetic and the temperature of the gas increases. At high temperatures, the gas turns into a plasma.What happens to gas when temperature increases?
The volume of the gas increases as the temperature increases. As temperature increases, the molecules of the gas have more kinetic energy. They strike the surface of the container with more force. If the container can expand, then the volume increases until the pressure returns to its original value.How does temperature affect gas volume?
Charles' law (Gay-Lussac's law) This law states that the volume and temperature of a gas have a direct relationship: As temperature increases, volume increases, when pressure is held constant. Heating a gas increases the kinetic energy of the particles, causing the gas to expand.What does buoyancy depend on?
The buoyancy force experienced by an object depends on its shape. The fraction of an object's volume that's submerged is given by the ratio of its average density to that of the fluid: ¯ρobj/ρfl ρ ¯ obj / ρ fl . An object floats if the buoyancy force exerted on it by the fluid balances its weight.How does heat affect pressure?
In an open space increased heat will cause the the molecules to more faster allowing them to travel farther from each other or expand simply by increasing their volumes. The force that the molecules are pressing against the enclosure is called pressure so temperature is directly proportional to pressure.What happens when you heat a balloon?
In summary: as you heat a gas, it expands. In a balloon, cooling the gas inside would cause the circumference to decrease. Answer 3: If a balloon is heated up, the gas inside will expand, causing the circumference of the balloon to increase.How do you heat up a balloon?
The air inside will have a pressure exactly equal to ambient pressure, because the elastic is not being stretched. Now cover the balloon completely in warm water for a few minutes, and it should inflate slightly. Finally, remove the balloon from the water and quickly perforate it with a sharp object.How do you increase pressure in a balloon?
As the balloon volume decreases, pressure inside the balloon increases. It eventually is able to balance the outside air pressure. You end up with a balloon filled with cold high density air. If you warm air it will expand and density will decrease until the pressure inside and outside the parcel are equal.What is the air inside the balloon?
A balloon is a flexible bag that can be inflated with a gas, such as helium, hydrogen, nitrous oxide, oxygen, air. For special tasks, balloons can be filled with smoke, liquid water, granular media (e.g. sand, flour or rice), or light sources.How does heat affect the volume of gas in a balloon?
The particles of the gas in the balloons moved farther away from each other when the temperature rose, resulting in a higher volume. The particles came closer together when the temperature dropped, resulting in a smaller volume. The higher the temperature gets, the more the volume of the gas in a balloon will increase.What is the pressure inside the balloon?
If it is filled with air, it is about 950 millibars plus or minus 75 millibars. Ok, that was fun. Seriously, it is only slightly higher than atmospheric pressure. That is 14.7 PSI more or less.How does pressure affect a balloon?
The higher up the balloon goes the less air there is to push down on the balloon so the pressure decreases. The outside air pressure is pushing on the balloon less than it was when the balloon was at sea level. Therefore, the balloon will expand since there is less pressure being applied on it.Is hot or cold air lighter?
The less dense hot air then floats in the more dense cold air much like wood floats on water because wood is less dense than water. Warm air is lighter than cold air. Because the cold air contains some of the molecules of water. So warm air is lighter than cold air.Is pressure inside a balloon greater?
The pressure inside the balloon is higher than the pressure on the outside. If the pressure was same then there would be no airflow and if the pressure outside was higher , air will flow from outside to the inside of the balloon. That makes is obvious that the inside of the balloon is at a higher pressure.What happens to the pressure of a gas when it is heated at constant volume?
The Pressure Law. The pressure law states: "For a fixed mass of gas, at a constant volume, the pressure (p) is directly proportional to the absolute temperature (T)." Heat energy is applied to the cylinder and the temperature of the gas increases.What type of gas is used in hot air balloons?
helium