Why does molar mass equal atomic mass?
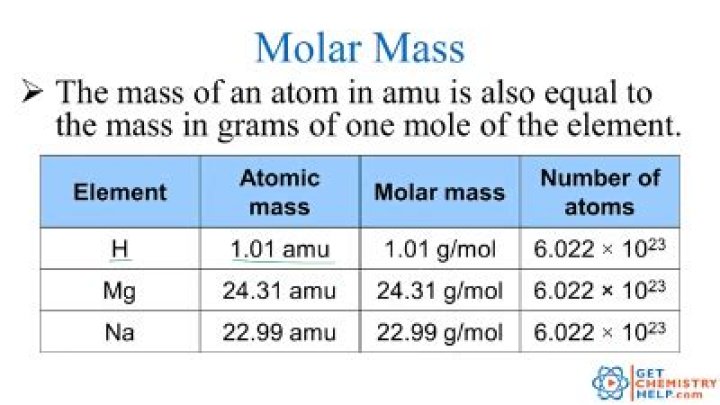
.
Also, why are atomic mass and molar mass the same?
Explanation: Atomic mass is the mass of an atom and is given in a.m.u. (atomic mass unit). However, a molar mass is the mass of one mole atoms or molecules and is given in grams. one mole is equal to Avogadro's number ( NA=6.022×1023 ) atoms or molecules.
Subsequently, question is, why is the atomic mass in a fraction? The atomic masses of most elements are fractional because they exist as a mixture of isotopes of different masses. Most elements occur as a mixture of isotopes of different masses. The fractional atomic masses arise because of this mixture. mass = total mass of all atoms/number of atoms.
Likewise, people ask, is molar mass the same as mass number?
Molar mass is the mass of one mole - that's 6.022 x 10 - of atoms, or molecules. When molar mass is given in grams, it has the same numerical value as relative atomic (or molecular) mass. The two can be used interchangeably in most applications, although there is a subtle difference in their meanings.
What has a mass of 1 amu?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.
Related Question AnswersWhat is atomic mass measured in?
The atomic mass of an element is the average mass of the atoms of an element measured in atomic mass unit (amu, also known as daltons, D). The atomic mass is a weighted average of all of the isotopes of that element, in which the mass of each isotope is multiplied by the abundance of that particular isotope.What is atomic mass and molecular mass?
The atomic mass unit (u) is a unit that describes the masses of individual atoms and molecules. The atomic mass is the weighted average of the masses of all isotopes of an element. The molecular mass is the sum of the masses of the atoms in a molecule.Is atomic mass and molecular weight the same?
Molecular weight is the mass of a molecule of a substance. It can also be called molecular mass. The units for molecular weight are atomic mass units (amu). Molar mass is the mass of one mole of a substance.What is the formula for molar mass?
Calculating Molar Mass Molar mass is the mass of a given substance divided by the amount of that substance, measured in g/mol. For example, the atomic mass of titanium is 47.88 amu or 47.88 g/mol.What is atomic mass in science?
Atomic mass or weight is the average mass of the protons, neutrons, and electrons in an element's atoms.What is difference between AMU and Gram?
The main difference between amu and grams is that amu is used to express the mass in atomic level whereas gram is used as a metric unit of mass.What is difference between atomic mass and gram atomic mass?
Answer: When atomic mass is defined in terms of gram it is known as gram atomic mass. When molecular mass is defined in terms of gram it is known as gram molecular mass. Molecular mass in gram is equal to atomic weight of the atom.How many moles are in a gram?
We assume you are converting between moles In and gram. You can view more details on each measurement unit: molecular weight of In or grams The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles In, or 114.818 grams.How do you convert mass to Grams?
To convert from g into units in the left column divide by the value in the right column or, multiply by the reciprocal, 1/x. To convert among any units in the left column, say from A to B, you can multiply by the factor for A to convert A into grams then divide by the factor for B to convert out of grams.How do you convert molar mass to Grams?
There are three steps to converting moles of a substance to grams:- Determine how many moles are given in the problem.
- Calculate the molar mass of the substance.
- Multiply step one by step two.



