Why does Benedict's solution need to be heated?
.
Also to know is, what happens when you heat Benedict's solution?
When Benedict's solution and simple carbohydrates are heated, the solution changes to orange red/ brick red. This reaction is caused by the reducing property of simple carbohydrates. The copper (II) ions in the Benedict's solution are reduced to Copper (I) ions, which causes the color change.
Beside above, what temperature does Benedict's work at? 95 °C
Likewise, people ask, why do the colors intensify with the continued application of heat?
Answer: because the colour changes from the initial blue copper sulphate to green to red as more reaction takes place and more red copper oxide is formed.
What happens to the reagent when it is heated and sugars are present?
As it is heated in the presence of reducing sugars, it turns yellow to orange. The "hotter" the final color of the reagent, the higher the concentration of reducing sugar.
Related Question AnswersWhy does sucrose not react with Benedict's solution?
Because Sucrose (table sugar) contains two sugars (fructose and glucose) joined by their glycosidic bond in such a way as to prevent the glucose isomerizing to aldehyde, or the fructose to alpha-hydroxy-ketone form. Sucrose is thus a non-reducing sugar which does not react with Benedict's reagent.How do you make Benedict's solution?
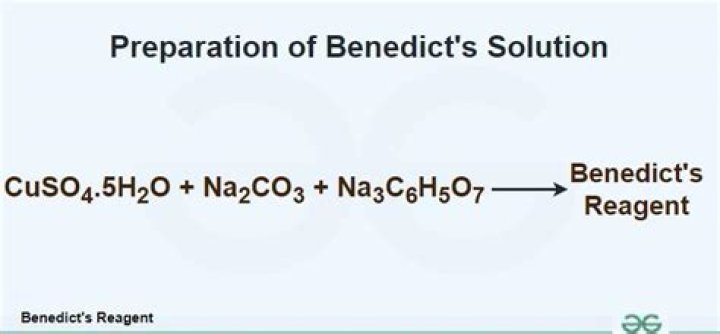
One litre of Benedict's reagent can be prepared by mixing 17.3 grams of copper sulfate pentahydrate (CuSO4. 5H2O), 100 grams of sodium carbonate (Na2CO3), and 173 grams of sodium citrate in distilled water (required quantity).Is Benedict's solution toxic?
OSHA: No component of this product present at levels greater than or equal to 0.1% is identified as a carcinogen or potential carcinogen by OSHA. May be harmful if inhaled. Causes respiratory tract irritation. Ingestion Toxic if swallowed.How does Benedict's solution react with sugar?
The principle of Benedict's test is that when reducing sugars are heated in the presence of an alkali they get converted to powerful reducing species known as enediols. When Benedict's reagent solution and reducing sugars are heated together, the solution changes its colour to orange-red/ brick red.Is Benedict's test qualitative or quantitative?
Is the Benedict's Test for reducing sugars qualitative or quantitative? The test may be qualitative, or it may be quantitative. The qualitative test produces a colour change from blue to green to yellow to orange to brick red.What is Benedict's solution made of?
Benedict's solution (Fehling's solution) is used to test for simple sugars such as glucose. It is a clear blue solution which is a combination of copper sulfate, sodium citrate, and sodium carbonate.What Colour does Benedict's turn when glucose is present?
Benedict's solution is used to test for simple sugars, such as glucose. It is a clear blue solution of sodium and copper salts. In the presence of simple sugars, the blue solution changes color to green, yellow, and brick-red, depending on the amount of sugar.Is starch a reducing sugar?
Glucose has a free aldehyde group which can be oxidized to the acidic groups. Hence, glucose is a reducing sugar. Starch and Cellulose are polysaccharides. The glucose in starch and cellulose does not contain a free aldehyde group and hence, starch and cellulose do not act as reducing sugars.Why does the Benedict's test work?
Benedict's reagent is made by complexing Cu+2 (from Copper sulfate) ions with citric acid in a basic medium(Sodium Carbonate. Benedict's Reagent is used to detect reducing sugars. When this happens the color of the reagent turns from blue to colorless by the reduction of copper(II) to Copper(I) that is colorless.Why is Benedict's reagent more stable than Fehling's reagent?
why is Benedict reagent more stable than fehling's reagent? Fehling's solution is made up out of two separate solutions, is caustic, and doesn't keep well. Benedict's solution is more stable, is a single solution, and has no caustic properties, making it easier to handle.Why does some food need to be crushed?
Digestion is enhanced by the breaking of food into small particles that expose more food surface to the action of digestive enzymes; the more food surface exposed, the more efficient the process of digestion.Is lactose a reducing sugar?
Lactose is composed of a molecule of galactose joined to a molecule of glucose by a β-1,4-glycosidic linkage. It is a reducing sugar that is found in milk.How do you test for fat?
Test for Fats- Take a small quantity of the food item to be tested.
- Wrap the food item in a piece of paper and crush it.
- Straighten the paper.
- Dry the paper by keeping it in sunlight for a while.
- Observe the paper.
- An oily patch on the paper indicates the presence of fats in the tested food item.



