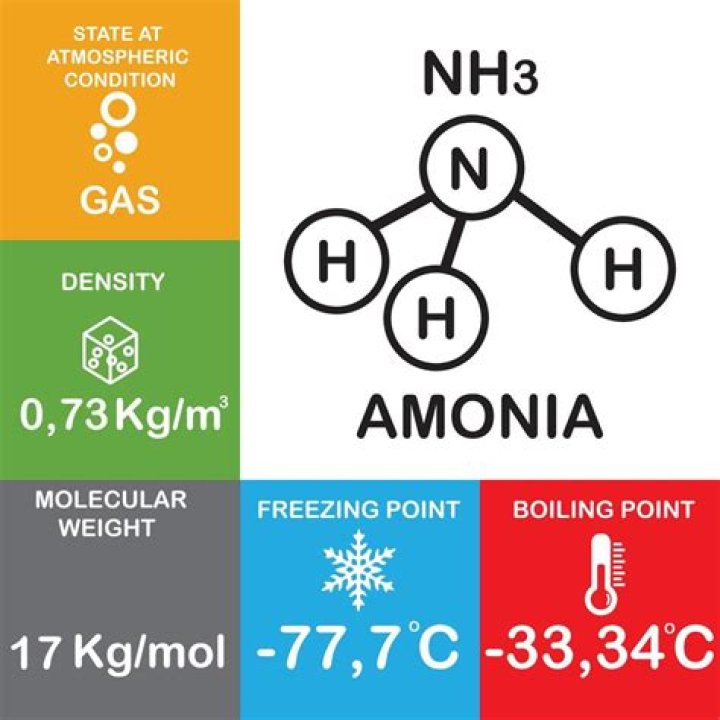
Ammonia, NH Because the nitrogen is only forming 3 bonds, one of the pairs must be a lone pair. The electron pairs arrange themselves in a tetrahedral fashion as in methane. That forces the bonding pairs together slightly - reducing the bond angle from 109.5° to 107°..
Beside this, why does nh3 have 107 bond angle?
NH3 Bond Angles In NH3, the bond angles are 107 degrees. It is close to the tetrahedral angle which is 109.5 degrees. But it is 107 degrees because the bonding pair occupies less space than the nonbonding pair.
Similarly, why bond angle of nh3 is greater than h2o? as H2O has two lone pairs so It repels the bond pairs much more and makes bond angle shorter of 104.5 degrees and as NH3 has one lone pair that repels the three bond pair but not much effectively and strongly as two lone pairs of water repel the bond pair so the bond angle between hydrogen atom of ammonia is 107.5
Also to know is, what is the bond angle in ammonia?
The bond angle in a molecule of ammonia (NH3) is 107 degrees so why, when part of a transition metal complex is the bond angle 109.5 degrees. This means that all four valence pairs are bonding and have therefore equal repulsive effects, meaning that the bond angles are equal at 109.5 degrees.
Why bond angle in water is less than that of ammonia?
Although geometries of NH3 and H2O molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. But in case of H2O, the O atom is surrounded by two bond pairs & TWO lone pair of electrons. Consequently ,the repulsion will be stronger & greater between the lone pairs & bond pairs.
Related Question Answers
What is the bond angle of h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.Which is the strongest bond?
Answer: Covalent bond is the strongest bond. Answer: There are a variety of ways atoms bond to one another.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Is nh3 bent or linear?
For example; four electron pairs are distributed in a tetrahedral shape. If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3). If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).What is the bond length of ammonia?
Each H is hydrogen bonded to a nearby nitrogen atom. Note that these hydrogen bonds (bond length 2.4 Å) are longer than normal ones (typical bond length ~2.0 Å), but they still contribute to the relatively high melting point (-78°C) of ammonia, compared to phosphine (PH3, melting point -133°C).Is bcl3 polar or nonpolar?
The molecular geometry of BCl3 is trigonal planar with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.What are the expected bond angles in ICl4+?
What are the expected bond angles of ICl4+? Choose all that apply: a) 90 degrees b)109.5 degrees c)120 degrees d)180 degrees.Which bond is stronger sigma or pi why?
To elaborate further on Sam Yan's answer, sigma bonds are stronger than pi bonds because the atomic orbitals forming sigma bonds overlap to a greater extent than the orbitals forming pi bonds. Sigma bonds are formed by head-on overlaps, while pi bonds are formed by side-on overlaps.Why is so2 bent and not linear?
CO2 is linear and the SO2 structure is bent, because CO2 has a negative oxygen on each side of the positive carbon they cancel each other out. In the SO2 structure the oxygen are not in lined with each other which means there a positive and negative end.How many lone pairs are in nh3?
How many bond pairs of electrons are found in NH3 molecules? NH3 molecules have 3 bonded pairs of electrons. It is SP3 hybridised.. So have 3 bond pairs with 3 H atom and one lone pair.What is the Lewis structure of ammonia?
The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons.What is the shape of nh4+?
In NH4+ion four bond pairs are present. The repulsion between bond pairs is less as compared to the bond pair-lone pair repulsion. Therefore the geometry is tetrahedral.Is ammonia polar or nonpolar?
Ammonia is a polar molecule: The trigonal pyramid geometry with the one lone electron pair contributes to the effect. The electrostatic potential clearly shows that the nitrogen is partially negative whereas the hydrogens are partially positive.Is h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.Is nh3 tetrahedral or trigonal pyramidal?
Be sure to use the number of available valence electrons you found earlier. Hydrogen (H) atoms always go on the outside of a Lewis structure. Originally Answered: What is the NH3 molecule shape? NH3 (Ammonia) electron geometry is “Tetrahedral” but its molecular geometry is “Trigonal Pyramidal”.Does nh3 have a dipole moment?
NH3 has a dipole moment while BF3 does not. By the VSEPR theory, the shape of the NH3 molecule turns out to be tetrahedral with 3 bond pairs and 1 lone pair. Thus, there is no chance of cancellation of the dipole vector as the vectors are inclined at an angle of 107°.What is the bond angle of c2h4?
The C2H4 bond angle will be about 120 degrees since it has a trigonal planar molecular geometry.What is the bond angle of bcl3?
120
oWhat is the bond angle of CCl4?
1 Answer. CCl4 has a tetrahedral geometry with bond angles of 109.5 °.