The antibody recognizes a unique part of the foreign target, called an antigen. Each tip of the “Y” of an antibody contains a paratope that is specific for one particular epitope (analogous to a lock and key) on an antigen, allowing these two structures to bind together with precision..
Thereof, what happens after antibodies bind a specific antigen?
When some antibodies combine with antigens, they activate a cascade of nine proteins, known as complement, that have been circulating in inactive form in the blood. Complement forms a partnership with antibodies, once they have reacted with antigen, to help destroy foreign invaders and remove them from the body.
Similarly, how do antibodies recognize specific antigens? Immunogens can be designed so that antibodies are generated against specific proteins. The specific region on an antigen that an antibody recognizes and binds to is called the epitope, or antigenic determinant. An epitope is usually 5-8 amino acids long on the surface of the protein.
Beside this, what is the relationship between antigens and antibodies?
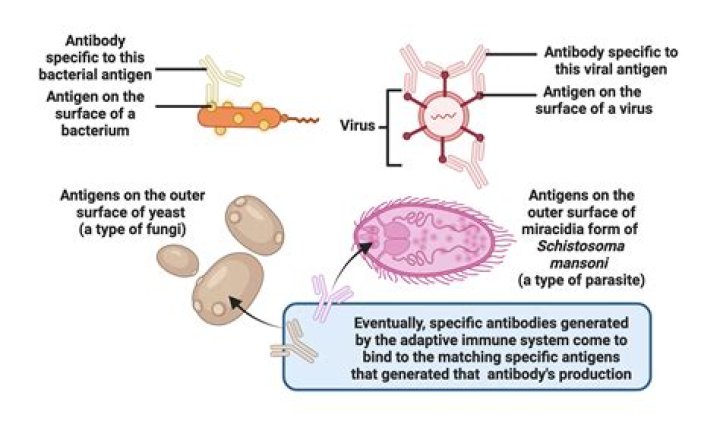
Antigens are molecules capable of stimulating an immune response. Each antigen has distinct surface features, or epitopes, resulting in specific responses. Antibodies (immunoglobins) are Y-shaped proteins produced by B cells of the immune system in response to exposure to antigens.
How does the structure of an antibody allow it to bind to a specific antigen?
Each antibody consists of four polypeptides– two heavy chains and two light chains joined to form a "Y" shaped molecule. This variable region, composed of 110-130 amino acids, give the antibody its specificity for binding antigen.
Related Question Answers
Do antibodies destroy antigens?
Antigens are proteins that are found on the surface of the pathogen. The antibodies destroy the antigen (pathogen) which is then engulfed and digested by macrophages. White blood cells can also produce chemicals called antitoxins which destroy the toxins (poisons) some bacteria produce when they have invaded the body.How many types of antigen and antibody are found?
With the help of this binding, the antigens are eliminated from the body. This occurs either through direct neutralization or with the help of tagging of other arms of the immune system. There are five kinds of antibodies, such as Immunoglobulins M, G, E, D and A.How do antibodies function?
Antibodies have three main functions: 1) Antibodies are secreted into the blood and mucosa, where they bind to and inactivate foreign substances such as pathogens and toxins (neutralization). 2) Antibodies activate the complement system to destroy bacterial cells by lysis (punching holes in the cell wall).Where do antigens bind on antibodies?
The paratope is the part of an antibody which recognizes an antigen, the antigen-binding site of an antibody. It is a small region (15–22 amino acids) of the antibody's Fv region and contains parts of the antibody's heavy and light chains. The part of the antigen to which the paratope binds is called an epitope.What is the function of the binding site in an antibody?
Antigen-binding site. (A) The hinge region of an antibody molecule opens and closes to allow better binding between the antibody and antigenic determinants on the surface of an antigen. (B) Hinge flexibility also facilitates the cross-linking of antigens into large antigen-antibody complexes.What are the factors affecting antigen antibody reaction?
Avidity is perhaps a more informative measure of the overall stability or strength of the antibody-antigen complex. It is controlled by three major factors: antibody epitope affinity, the valence of both the antigen and antibody, and the structural arrangement of the interacting parts.What are the mechanisms of antibody action?
Mechanisms of antibody action: Antibodies may inhibit infection by (a) preventing the antigen from binding to its target, (b) tagging a pathogen for destruction by macrophages or neutrophils, or (c) activating the complement cascade.What are different types of antibodies?
There are five different antibody isotypes seen in humans: IgG, IgA, IgM, IgE, and IgD. IgG is the antibody isotype that most people think of when they're talking about antibodies. It is the antibody that is built by immunization. It activates an immune cascade that can eliminate some forms of infection.What are antigens and antibodies in blood?
Antigens And Antibodies. Antibodies are a specific type of immune-system proteins known as immunoglobulins, whose role is to fight infections by binding themselves to antigens. In the case of the ABO blood groups, the antigens are present on the surface of the red blood cell, while the antibodies are in the serum.What is the relationship between antigens and antibodies quizlet?
What is the relationship between antigens and antibodies? Antigens are any molecules that elicit adaptive immune responses. Antigens protrude from pathogens or other intruding substances. Antibodies are proteins found in blood plasma that attach to one particular kind of antigen and help counter its effects.Are antigens bad?
The immune system protects the body from possibly harmful substances by recognizing and responding to antigens. Antigens are substances (usually proteins) on the surface of cells, viruses, fungi, or bacteria. Your immune system learns to see these antigens as normal and usually does not react against them.What are some examples of antigens?
Foreign antigens originate from outside the body. Examples include parts of or substances produced by viruses or microorganisms (such as bacteria and protozoa), as well as substances in snake venom, certain proteins in foods, and components of serum and red blood cells from other individuals.What causes high levels of antibodies in blood?
If you have high levels of antithyroglobulin antibodies in your blood, it may be a sign of serious autoimmune disorder, such as Graves' disease or Hashimoto thyroiditis. If you test positive for these antibodies, and your doctor can't identify an underlying cause, they may monitor you for emerging health problems.What happens when antigen binds to antibody?
When some antibodies combine with antigens, they activate a cascade of nine proteins, known as complement, that have been circulating in inactive form in the blood. Complement forms a partnership with antibodies, once they have reacted with antigen, to help destroy foreign invaders and remove them from the body.How do antigens and antibodies work?
Antigens are molecules capable of stimulating an immune response. Each antigen has distinct surface features, or epitopes, resulting in specific responses. Antibodies (immunoglobins) are Y-shaped proteins produced by B cells of the immune system in response to exposure to antigens.How do T cells recognize an antigen?
Antigen recognition by T cells. T cells can detect the presence of an intracellular pathogen because infected cells display on their surface peptide fragments derived from the pathogen's proteins. These foreign peptides are delivered to the cell surface by specialized host-cell glycoproteins.What is the structure of an antibody?
Introduction. Antibodies are immune system-related proteins called immunoglobulins. Each antibody consists of four polypeptides– two heavy chains and two light chains joined to form a "Y" shaped molecule.Why do you need to assay positive and negative control samples?
Why do you need to assay positive and negative control samples as well as your experimental samples? Controls are needed to make sure the assay is working correctly. Conversely, without a negative control, there is no way of knowing if all samples (positive or not) would have given a positive result.