Why do alkenes show electrophilic addition reaction?
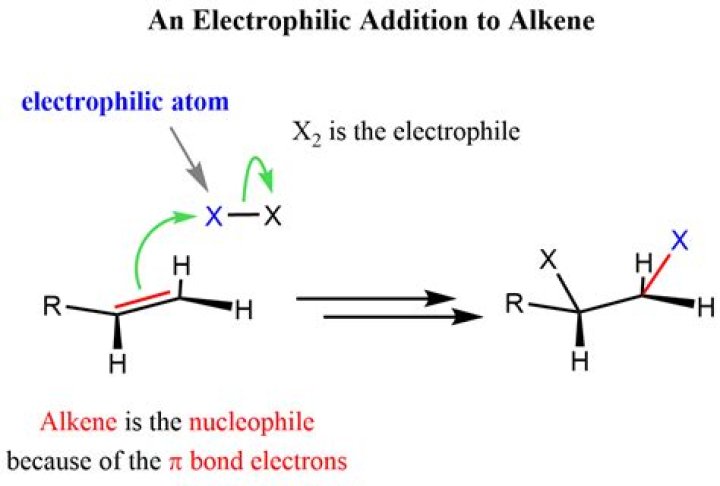
.
Herein, why do alkenes undergo electrophilic addition reaction?
Alkenes usually undergo electrophilic addition reaction at carbon-carbon double bond. Alkenes have loosely held pi electrons, a source for the electrophilic attack, which is correct Reason. The electrophilic attack leads to a carbocation intermediate which is a consequence of the Assertion.
Secondly, what is the addition reaction of alkenes? Addition reactions are when two smaller substances come together to form a larger substance. For example, the reaction of bromine (HBr) and propene (C_3H_6) is an addition reaction. In this case an electrophile breaks up the double bond, thus allowing the carbon to bond new a new atom or atoms.
Similarly, what is an electrophilic addition reaction examples?
The reactions are examples of electrophilic addition. Hydrogen chloride and the other hydrogen halides add on in exactly the same way. For example, hydrogen chloride adds to ethene to make chloroethane: The only difference is in how fast the reactions happen with the different hydrogen halides.
What does electrophilic addition Mean?
Electrophilic addition is a reaction between an electrophile and nucleophile, adding to double or triple bonds. An electrophile is defined by a molecule with a tendency to react with other molecules containing a donatable pair of electrons.
Related Question AnswersWhat are the conditions for electrophilic addition?
In organic chemistry, an electrophilic addition reaction is an addition reaction where, in a chemical compound, a π bond is broken and two new σ bonds are formed. The substrate of an electrophilic addition reaction must have a double bond or triple bond.Why does electrophilic addition occur?
An electrophilic addition reaction is an addition reaction which happens because what we think of as the "important" molecule is attacked by an electrophile. The "important" molecule has a region of high electron density which is attacked by something carrying some degree of positive charge.Which is more reactive towards electrophilic addition reaction?
Alkynes are less reactive than alkenes towards electrophilic addition reaction. As a result the reactivity decrease. The pie-electron cloud of alkyne makes a cylindrical electron cloud around the triple bond . Makes the electrons more delocalized and stable.What is the difference between nucleophilic and electrophilic addition?
Summary – Nucleophilic vs Electrophilic Addition The key difference between nucleophilic and electrophilic addition is that in nucleophilic addition reactions, an electron-rich component is added to a molecule, whereas in electrophilic addition, an electron-deficient species is added to a molecule.Which compound will be most reactive for electrophilic addition reaction?
In series of activating group O H OH OH comes first then O C H 3 OCH_3 OCH3, hence, phenol is most reactive towards electrophilic substitution reaction.Is hydrogenation an electrophilic addition reaction?
Hydrogenation. There is another reaction of alkenes, hydrogenation, which deserves mention but which is not related to the electrophilic addition mechanism. Hydrogenation is the addition of molecular hydrogen (H22) to the alkene double bond. This converts a simple alkene into an alkane.Is a double bond electrophilic or nucleophilic?
In organic chemistry, a nucleophilic addition reaction is an addition reaction where a chemical compound with an electron-deficient or electrophilic double or triple bond, a π bond, reacts with electron-rich reactant, termed a nucleophile, with disappearance of the double bond and creation of two new single, or σ,Is h2o a nucleophile?
Yes, water is a nucleophile. Water is both a nucleophile and an electrophile. and as electrophile by giving a proton to a nucleophile.Is alkene nucleophile or electrophile?
Yes, alkenes are nucleophiles. The π bond is localized above and below the C-C σ bond. These π elecrons are relatively far from the nuclei and are loosely bound. An electrophile can attract those electrons and pull them away to form a new bond.What do you mean by addition reaction?
An addition reaction, in organic chemistry, is in its simplest terms an organic reaction where two or more molecules combine to form a larger one (the adduct). There are two main types of polar addition reactions: electrophilic addition and nucleophilic addition.Is water an electrophile?
Water: The oxygen atom of water has two lone pairs and a d- charge (oxygen is more electronegative than hydrogen). This suggests that water can behave an a nucleophile. Each hydrogen atom bears a d+ charge, so the molecule can behave as an electrophile as well. Many molecules can be both nucleophiles and electrophiles.What is meant by an electrophile?
In organic chemistry, an electrophile is an electron pair acceptor. Electrophiles are positively charged or neutral species having vacant orbitals that are attracted to an electron rich centre. It participates in a chemical reaction by accepting an electron pair in order to bond to a nucleophile.Is HBr a nucleophile or electrophile?
Re: HBr electrophile or nucleophile yes. the Br is the nucleophile because is is slightly negative, pulling the electrons in the shared bond with Hydrogen slightly more toward it, making Br electron rich. This leaves H slightly positive and therefore an electrophile as it is being deprived of electrons slightly.What type of reaction is bromination?
Bromination: Any reaction or process in which bromine (and no other elements) are introduced into a molecule. Bromination of an alkene by electrophilic addition of Br2. Bromination of a benzene ring by electrophilic aromatic substitution. Bromination of a benzylic position by a free radical substitution reaction.What are the two steps in a nucleophilic addition mechanism?
What are the two steps in a nucleophilic addition mechanism? A. Nucleophilic attack followed by protonation.What are the types of addition reaction?
We will study three main types of reactions - addition, elimination and substitution.- An addition reaction occurs when two or more reactants combine to form a single product.
- An elimination reaction occurs when a reactant is broken up into two products.



