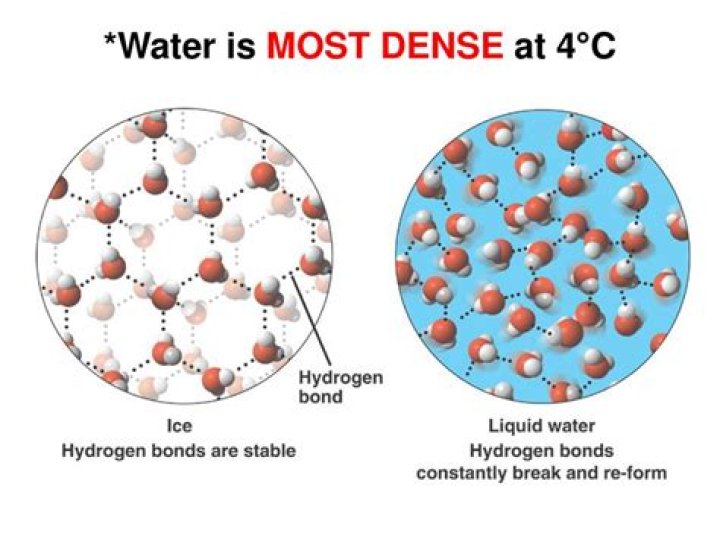
Water is densest at 3.98°C and is least dense at 0°C (freezing point). Water density changes with temperature and salinity. When water freezes at 0°C, a rigid open lattice (like a web) of hydrogen-bonded molecules is formed. It is this open structure that makes ice less dense than liquid water..
Also to know is, which state of water has the greatest density?
Water has a higher density in the liquid state than the solid, so ice cubes float.
Secondly, how dense can water get? 997 kg/m³
Similarly, you may ask, which form of water is least dense?
Answer and Explanation: The type of water that is least dense is water vapor. Water vapor is the gas form of water, where the molecules of water have very little bonds
Why is water most dense at 4c?
At 4 degrees C these two forces work out to make water the most dense. That is: the thermal properties are not enough to break all the h-bonds apart, but the h-bonds have not formed enough to widen the distance between water molecules to be as great as in ice (which is why ice is lighter than water).
Related Question Answers
Is density a chemical property?
The general properties of matter such as color, density, hardness, are examples of physical properties. Properties that describe how a substance changes into a completely different substance are called chemical properties. Flammability and corrosion/oxidation resistance are examples of chemical properties.Is ice more dense than water?
When water freezes, water molecules form a crystalline structure maintained by hydrogen bonding. Solid water, or ice, is less dense than liquid water. Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart, which lowers the density.What is highest density?
As it turns out, either of two elements can be considered the element with the highest density: osmium or iridium. At room temperature and pressure, the calculated density of osmium is 22.61 g/cm3 and the calculated density of iridium is 22.65 g/cm3.Which matter has highest density?
Solids
What is the density of pure water?
A: Pure water has a density of 0.99823 grams/cubic centimeter at 1 atm pressure and a temperature of 20 degrees Celsius (68 degrees Fahrenheit).How do you find the density?
To find the density of any object, you need to know the Mass (grams) of the object, and its Volume (measured in mL or cm³). Divide the mass by the volume in order to get an object's Density.Does density increase with temperature?
Density changes with temperature because volume changes with temperature. Density is mass divided by volume. As you heat something up, the volume usually increases because the faster moving molecules are further apart. Since volume is in the denominator, increasing the volume decreases the density.Is a gas or liquid more dense?
Consequently, liquids are much denser than gases. It is THEORETICALLY POSSIBLE TO HAVE A GAS HAVING DENSITY MORE THAN THAT OF THE LIQUID OF THE SAME SUBSTANCE. In general, liquids are denser than gases; solids are denser than liquids.Can you change the density of water?
As water drops below that temperature, it becomes less dense, which is why ice floats. However, temperature fluctuates naturally, so if you wish to increase the density permanently, you can add salt to the water. This increases the water's mass without increasing its volume. Thus, its density increases.Why is water called the universal solvent?
Water is capable of dissolving a variety of different substances, which is why it is such a good solvent. And, water is called the "universal solvent" because it dissolves more substances than any other liquid. This allows the water molecule to become attracted to many other different types of molecules.How dense is milk?
1033 kg /m3
Is water Polar?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.Why is water's density important?
Water's Density Makes Life Possible Most things become more dense when they cool because their molecules are condensing together, and become less dense when they heat up because their molecules are moving farther apart from each other.What affects the density of water?
There are two main factors that make ocean water more or less dense than about 1027 kg/m3: the temperature of the water and the salinity of the water. Ocean water gets more dense as temperature goes down. So, the colder the water, the more dense it is. Increasing salinity also increases the density of sea water.Is water denser than oil?
Oil is more dense than alcohol, but less dense than water. The molecules that make up the oil are larger than those that that make up water, so they cannot pack as tightly together as the water molecules can. They take up more space per unit area and are less dense.How dense is ice?
The density of ice is about 0.92 g cm-3 and that of water is about 1.00 g cm-3 at 0 °C. Cooking oil has a density between these two and therefore ice floats on the oil whereas water sinks. Most solids are denser than their liquids.What liquid is heavier than water?
Mercury is a liquid at room temperature and has a density of 13.6 times that of water (careful, mercury's poisonous). Most metals, when melted, will be heavier than water (but may be hotter than water will tolerate), such as molten lead or steel (lead's also poisonous!).What temp is water most dense?
As water cools it becomes more dense. At 39°F (or 3.98°C to be exact) water is the most dense. This is because the molecules are closest together at this temperature.Does water float on water?
As water warms, it expands, decreasing density. As salt concentration rises, density increases, because the salt molecules can occupy spaces between the water molecules. Denser water sinks beneath water that is less dense. You can see for yourself if water can float on water.