Hard bases are highly electronegative and of low polarizability. Examples of Hard Bases: F-, OH-, NH3, N2H4, ROH, H2O, SO42-, PO43- Hard bases react more readily to form stable compounds and complexes with hard acids..
Considering this, what are hard acids and bases give examples?
Metal–ligand interaction is an example of a Lewis acid–base interaction. Lewis bases can be divided into two categories: hard bases contain small, relatively nonpolarizable donor atoms (such as N, O, and F), and.
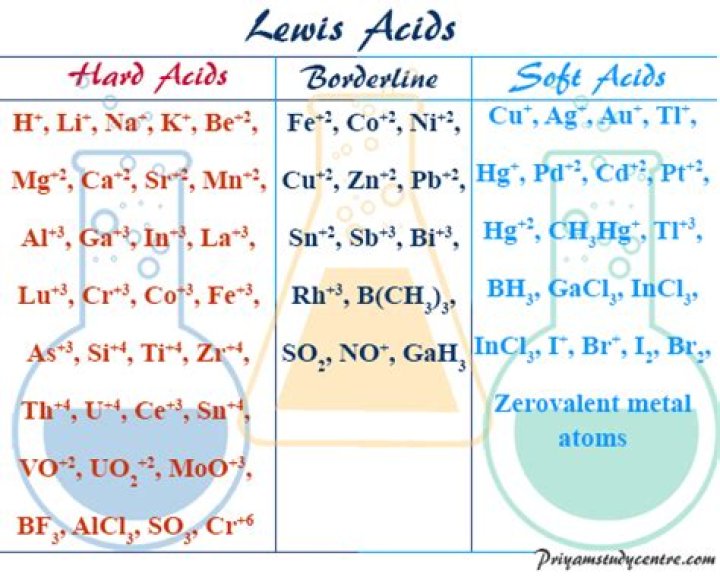
Hard and Soft Acids and Bases.
| Acids | Bases |
| hard | Al3+, Sc3+, Cr3+ | CO32− |
| Ti4+ | PO43− |
| soft | BF3, Al2Cl6, CO2, SO3 |
| Cu+, Ag+, Au+, Tl+, Hg22+ | H− |
why do hard acids prefer hard bases? The theory elaborates that hard acids prefer to bond with hard bases, and the resulting adduct tend to have more ionic character in its bonding. Correspondingly, soft acids prefer to bond with soft bases, and their adducts are more covalent in nature.
Furthermore, is oxygen a hard or soft base?
This observation has eventually led to a classification system called Hard and Soft Acids and Bases (HSAB). In a nutshell, smaller or more highly charged metal ions are called hard acids. They are more likely to bind to hard bases, which typically have small donor atoms such as oxygen or nitrogen.
Is copper a hard or soft acid?
In this milieu, the principle of hard and soft acids and bases (HSAB) has been an extremely good guiding principle for the inorganic chemist. Copper(i) has been classified as a soft cation.
Related Question Answers
What are borderline acids and bases?
Borderline acids (in context, BA) are intermediate between hard and soft acids. Borderline bases (in context, BB) are intermediate between hard and soft bases. They tend to be larger and less electronegative than hard bases, smaller and more electronegative than soft bases.Why is CN a soft base?
Now, I know that the HOMO is the 5 σ MO and that the electron pair in the 5 σ MO is, to a large extent, more concentrated around the carbon. So, is CN− a soft base because it has a low energy HOMO but large magnitude HOMO coefficient?What is a soft Nucleophile?
Hard nucleophiles are usually the ones with a negative charge. They are also cylindrical or spear-shaped, allowing them to collide in an Sn2 reaction with the correct orientation. Soft nucleophiles are ones with a neutral charge and not such a penetrating shape, like water.Is sulfur soft or hard?
nitrogen tends to coordinate with harder acids such as Si, whereas the sulfur tends to coordinate with softer acids such as Pt2+. It is important to realize that hard/soft considerations have nothing to do with acid or base strength. An acid or a base may be hard or soft and also be either weak or strong.Is boron soft or hard?
Boron (B) is a soft, brown, amorphous or hard crystalline nonmetallic element (see nonmetal). It is also described as a metalloid element.What is soft base?
Soft Acids and Bases. Soft acids consist of large low charge cations and molecules with relatively high energy occupied molecular orbitals. Soft acids are readily polarizable. Examples of Soft Acids: Cs+, Cu+, Au+, Pt2+, Hg+, BH3, Br2, I2, RO+, quinones. Hard bases have low electronegative and low polarizability.Are metals hard or soft?
Hardness: All metals are hard except sodium and potassium, which are soft and can be cut with a knife. Valency: Metals have 1 to 3 electrons in the outermost shell of their atoms. Conduction: Metals are good conductors because they have free electrons.What is hard acid in chemistry?
Definition of Hard Acid. Hard acids are Lewis acids that are only weakly polarizable. Other things being approximately equal: hard acids react faster with hard bases and form stronger bonds with them. soft acids react faster with soft bases and form stronger bonds with them.What are hard and soft bases?
hard bases contain small, relatively nonpolarizable donor atoms (such as N, O, and F), and. soft bases contain larger, relatively polarizable donor atoms (such as P, S, and Cl).What metal is soft?
Cesium is considered the softest metal, Lead is also considered among the softest metals. Answer 3: Mercury is liquid (molten) at room temperature. Gallium, while solid (if soft) at room temperature, is liquid at body temperature.What are hard metals?
As a general rule, hard metals are made up of 90 - 94% tungsten carbide (reinforcement phase) or other carbides, which are very hard but also very brittle, and 6 - 10% cobalt, nickel or iron, which act as binding agents or so-called durability components.Is sulfur less polarizable than oxygen?
Sulfur is a larger atom than oxygen, making its electrons more polarizable. Thus, it is a stronger nucleophile than oxygen.Is sodium a soft metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.What is the strongest Lewis acid?
Boron tribromide is observed to be the strongest Lewis acid of these three compounds. The shorter boron-halogen bond distances in BF3 and BCl3 than in BBr3 are believed to lead to stronger halogen-to-boron p-p n bonding (see Section 5.10).What do you mean by Hsab?
HSAB concept is an initialism for "hard and soft (Lewis) acids and bases". Also known as the Pearson acid-base concept, HSAB is widely used in chemistry for explaining stability of compounds, reaction mechanisms and pathways. HSAB theory is also useful in predicting the products of metathesis reactions.Is calcium hard or soft?
Measures of water hardness General guidelines for classification of waters are: 0 to 60 mg/L (milligrams per liter) as calcium carbonate is classified as soft; 61 to 120 mg/L as moderately hard; 121 to 180 mg/L as hard; and more than 180 mg/L as very hard. What are soft ligands?
A ligand is essentially a Lewis base (since it donates an electron pair to the complex). Hard bases/ligands have a more concentrated, less polarizable electron pair. Examples are F- and HO-. Soft bases/ligands have a less concentrated, more polarizable electron pair. Examples are I- and HS-.Which bonding is involved in hard hard interaction?
ionic
Is carbon hard or soft?
Carbon, in various forms — called allotropes — runs the range from extremely soft (graphite) to extremely hard (diamond). It all comes down to the way the atoms are arranged. Hardness is described by Moh's scale of hardness, on which diamond is a “10”, the highest value, and graphite is no more than a “2”.