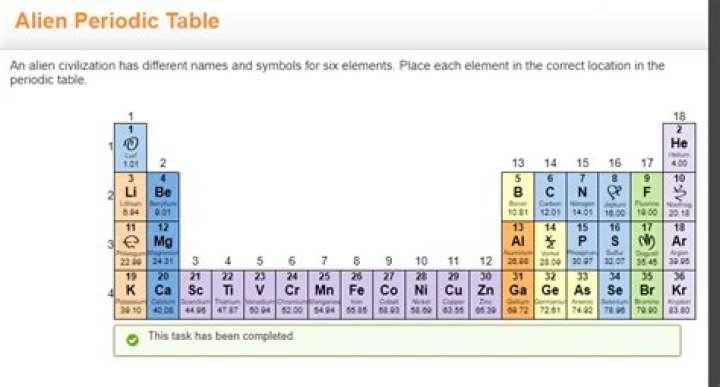
You will arrange the Aliens in some logical pattern so that they form an organized regular block, 8 groups and 5 periods. The resulting table is visually impressive and clearly shows the meaning of periodic trends. Each groups Alien Periodic Table will be different..
Also, what is the alien periodic table?
Since the alien scientists do not know the names of our elements, they have. radioed the following data on the known properties of the elements. Strangely, but luckily, there are no transition or rare earth elements on the alien. planet. This means that their periodic table consists on Iy of the"A" groups of elements.
Likewise, what are the groups in the periodic table? Group (periodic table)
- Group 1: the alkali metals (lithium family) *not including hydrogen.
- Group 2: the alkaline earth metals (beryllium family)
- Groups 3-12: the transition metals.
- Group 13: the triels (boron family)
- Group 14: the tetrels (carbon family)
- Group 15: the pnictogens (nitrogen family)
- Group 16: the chalcogens (oxygen family)
In this manner, what element is a metalloid by location?
The metalloids; boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po) and astatine (At) are the elements found along the step like line between metals and non-metals of the periodic table. Metalloids have properties of both metals and non-metals.
What is the metalloid with the lowest atomic mass?
Ernst is the metalloid with the lowest atomic mass.
Related Question Answers
What is the heaviest element?
uranium
What is the most metallic element on the planet?
francium
What is the most chemically active nonmetal on the planet?
The most chemically active nonmetal is apstrom. The lightest element on the planet is called pfsst (Pf), while the heaviest element is called elrado (El).How is the periodic table arranged?
The periodic table of elements arranges all of the known chemical elements in an informative array. Elements are arranged from left to right and top to bottom in order of increasing atomic number. Order generally coincides with increasing atomic mass. The rows are called periods.Which element has the greatest average atomic mass?
The atomic mass of an element is the summation of the number of neutrons and protons that are present in the element. Out of the options given, tellurium has the highest atomic mass which is 127.6.Is Rh a metalloid?
Nonmetals are (usually) poor conductors of heat and electricity, and are not malleable or ductile; many of the elemental nonmetals are gases at room temperature, while others are liquids and others are solids. The
metalloids are intermediate in their properties.
Metals, Metalloids, and Nonmetals.
What is the heaviest metalloid?
Metalloid
| Elements recognized as metalloids v t e |
| 13 | 14 |
| 2 | B Boron | C Carbon |
| 3 | Al Aluminium | Si Silicon |
| 4 | Ga Gallium | Ge Germanium |
What groups are metalloids?
What Are Metalloids? - Metalloids are the smallest class of elements, containing just six members: boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te).
- Q: Why does the chemical behavior of an element depend on its number of valence electrons?
What are the 8 metalloids?
The eight elements classified as metalloids are boron, silicon, germanium, arsenic, antimony, tellurium, astatine, and polonium. Metalloids occur along the diagonal stair-step between metals and non-metals.What is a metalloid element?
A metalloid is a chemical element that exhibits some properties of metals and some of nonmetals. Definition for metalloids: elements with properties intermediate between metals and nonmetals. Boron, silicon, germanium, arsenic, antimony, tellurium, and polonium are metalloids.Are metalloids magnetic?
A set of elements that have some properties of both metals and nonmetals and are found along a zigzag dividing line between metals and nonmetals on the periodic table; also known as metalloids. A force of attraction or repulsion between materials that produce a magnetic field.What is non metal in chemistry?
Non-metals are the elements in groups 14-16 of the periodic table. As opposed to metals, non-metallic elements are very brittle, and cannot be rolled into wires or pounded into sheets. The non-metals exist in two of the three states of matter at room temperature: gases (such as oxygen) and solids (such as carbon).Who discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.What elements are halogens?
Halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts).How many rows are in the periodic table?
seven
What is Group 7 on the periodic table called?
The Group 7 elements are called the halogens. They are placed in the vertical column, second from the right, in the periodic table . Chlorine, bromine and iodine are the three common Group 7 elements. Group 7 elements form salts when they react with metals. The term 'halogen' means 'salt former'.What are Group A elements?
Elements with similar reactivity are put into the same column or group. The elements in group IA are called the alkali metals. The elements in group IIA are called the alkaline earth metals. The elements in group VIIA are called the halogens and the elements in group VIIIA are called the noble gases or the inert gases.What is Group 2 on the periodic table called?
The alkaline earth metals are six chemical elements in group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).What are the 4 main groups on the periodic table?
The p-block elements may have more than one oxidation state, but when this happens, the most common oxidation states are separated by two units. Specific examples of main group elements include helium, lithium, boron, carbon, nitrogen, oxygen, fluorine, and neon.