Which elements in the actinide series are artificial?
.
Herein, which actinides are artificial?
The other actinides are purely synthetic elements. Nuclear weapons tests have released at least six actinides heavier than plutonium into the environment; analysis of debris from a 1952 hydrogen bomb explosion showed the presence of americium, curium, berkelium, californium, einsteinium and fermium.
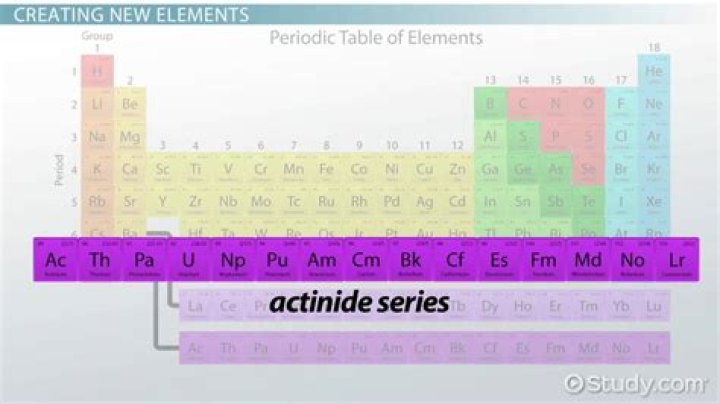
Likewise, which elements are lanthanides and actinides? The lanthanide and actinide series make up the inner transition metals. The lanthanide series includes elements 58 to 71, which fill their 4f sublevel progressively. The actinides are elements 89 to 103 and fill their 5f sublevel progressively.
Besides, how many elements are there in the actinide series in the periodic table?
14 elements
What is unique about actinide series?
Actinide Series of Metals The lanthanide series can be found naturally on Earth. Only one element in the series is radioactive. The actinide series is much different. They are all radioactive and some are not found in nature. Some of the elements with higher atomic numbers have only been made in labs.
Related Question AnswersAre actinides rare earth elements?
The Actinide series contains elements with atomic numbers 89 to 103 and is the third group in the periodic table. The series is the row below the Lanthanide series, which is located underneath the main body of the periodic table. Lanthanide and Actinide Series are both referred to as Rare Earth Metals.Can we live without Lanthanoids and Actinoids?
Yes, if you don't mind living in the Iron Age. None of the lanthanides or actinides (I'm old and use old-school terminology, OK?) are used in significant amounts in our bodies. A few, that are used, are used in minute amounts and probably can be substituted with other atoms.Which is the most reactive metal?
cesiumAre all actinides transuranium elements?
transuranic elements (transuranium elements) Elements with atomic numbers higher than that of uranium (92), the best known of which are members of the actinide series (atomic numbers 89 to 103). All transuranic elements are radioactive.Who discovered actinides?
The first actinides to be discovered were Uranium by Klaproth in 1789 and Thorium by Berezelius in 1829, but most of the Actinides were man-made products of the 20th century.Who discovered halogens?
Jöns Jacob BerzeliusAre halogens metals?
The halogens are a series of non-metal elements from group 17 of the periodic table (formerly VII). The halogens include fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The artificially created element 117 (ununseptium) may also be considered a halogen.Are actinides radioactive?
All actinides are radioactive and release energy upon radioactive decay; naturally occurring uranium and thorium, and synthetically produced plutonium are the most abundant actinides on Earth. These are used in nuclear reactors and nuclear weapons. The other actinides are purely synthetic elements.Which series of elements is mostly man made?
Actinides are the 15 elements with atomic numbers from 89 to 103. They are named after the first element in the series, actinium. The actinides group includes mostly man-made elements with only a few exceptions such as uranium and thorium.What are the actinide elements?
The usual list of elements in the actinide series is:- Actinium (Ac)
- Thorium (Th)
- Protactinium (Pa)
- Uranium (U)
- Neptunium (Np)
- Plutonium (Pu)
- Americium (Am)
- Curium (Cm)



