The chemical species isoelectronic to Ar are phosphide ion (P3− 3 − ), sulfide ion (S2− 2 − ), chloride ion (Cl− − ), potassium ion (K+ + ), calcium ion (Ca2+ 2 + ), and scandium ion (Sc3+ 3 + )..
Keeping this in view, which of the following ions is isoelectronic with an argon atom?
And we can see that the potassium ion, K+, has the same electronic configuration as the chloride ion, Cl-, and the same electronic configuration as an atom of argon, Ar. Therefore, Ar, Cl-, and K+ are said to be isoelectronic species.
Additionally, is ca2+ Isoelectronic with AR? Isoelectronic means that the entities (atoms, ions, molecules) in question have the same electronic structure and same number of valence electrons. For atoms/ions: Therefore, K+ and Ar are said to be isoelectronic. Ca2+ is also electronic with Ar, for example, and so is Cl-.
Besides, when forming an ion which element will be isoelectronic with argon AR?
To be isoelectronic with argon, which has 18 electrons, sulfur must gain two electrons. Therefore sulfur will form a 2- ion, becoming S2-.
What are isoelectronic elements?
Isoelectronic refers to two atoms, ions, or molecules that have the same electronic structure and the same number of valence electrons. The term means "equal electric" or "equal charge". Isoelectronic chemical species typically display similar chemical properties.
Related Question Answers
Which species is isoelectronic with Ne?
Answer and Explanation: Choices B and C are isoelectronic with neon. When the fluorine atom (atomic number 9) gains an electron it has 10 electrons, which is the sameHow do you know if a pair is Isoelectronic?
Isoelectronic molecules have the same electronic structure, and same number of valence electrons. If the number of electrons are the same, then they are isoelectronic. Ca2+ has 10 electrons. Hence they are isoelectronic.What element is isoelectronic with Krypton?
Expert Answers info Bromine with an atomic number of 35 acquires one electron to attain 36 electrons. This makes it isoelectronic with krypton.What is isoelectronic with mg2+?
Yes, the Mg2+ ion and the neutral neon atom are isoelectronic, which implies that they have the same number of electrons and, of course, the same electron configuration. The Mg2+ ion is formed when the neutral magnesium atom loses 2 electrons, which brings its total number of electrons to 10.What is calcium Isoelectronic with?
For an ion to be isoelectronic with an atom (or with another ion), it must have the same number of electrons surrounding its nucleus. Start with calcium, Ca , which has 20 electrons surrounding its nucleus. This implies that the Ca2+ cation will have 2 electrons less, or 18 electrons.Which noble gas is isoelectronic with a lithium ion?
Main group metals will typically form ions by losing enough electrons to become isoelectronic with the nearest noble gas. For example, lithium, whose configuration is [1s22s1], will typically lose one electron to become isoelectronic with helium, which has a configuration of [1s2] (see Figure above).Is argon a cation?
Each of the metals in Period 3 form cations by losing 1, 2, or 3 electrons, respectively. Each metal atom becomes isoelectronic with the preceding noble gas, neon. Anions form when an atom gains electrons and becomes negatively charged. Each nonmetal ion is isoelectronic with the following noble gas, argon.What is phosphorus Isoelectronic with?
And phosphorus forms a P3− ion. i.e. 18 electrons, and this is isoelectronic with neutral argon, i.e. Ar . And nitrogen forms a N3− ion. i.e. 10 electrons, and this is isoelectronic with neutral neon, i.e. Ne .Is NaCl Isoelectronic?
When cations and anions have a same number of electrons, they are called isoelectronic. ? they are isoelectronic. Sodium chloride is N a C l NaCl NaCl. Electrons in N a + Na^{+} Na+ is 10.Are NE and F Isoelectronic?
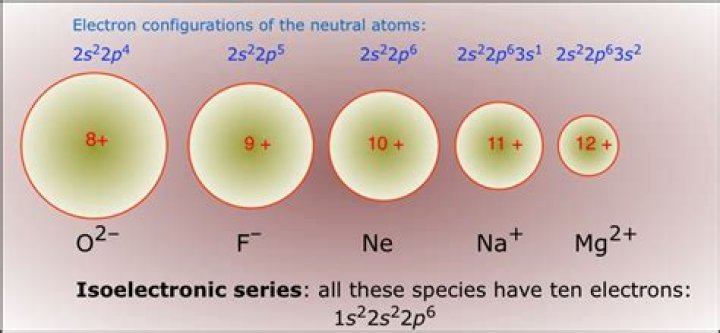
Atoms and ions that have the same electron configuration are said to be isoelectronic. Examples of isoelectronic species are N3–, O2–, F–, Ne, Na+, Mg2+, and Al3+ (1s22s22p6). For atoms or ions that are isoelectronic, the number of protons determines the size.What is argon used for?
It is used to fill incandescent and fluorescent light bulbs to prevent oxygen from corroding the hot filament. Argon is also used to form inert atmospheres for arc welding, growing semiconductor crystals and processes that require shielding from other atmospheric gases.Are Na+ and o2 Isoelectronic?
Are Na+ and O- isoelectronic? So Na+ ion and O2–ion are isoelectronic. Because both the ions have 10- electrons in them . ie., They have completed K and L- shell of electrons in them .Why is argon not reactive?
It is non-reactive because the shells are full. Argon has three electron shells. The third shell is filled with eight electrons. That is why it does not easily combine with other elements.How many core electrons are in AR?
Core = The argon core, [Ar], is composed of the core electrons in calcium. Argon contains 18 electrons, so calcium contains 18 core electrons. Note that the valence electrons are often all of those after the noble gas core, but not always.What noble gas is isoelectronic with sodium?
neon atom
How is argon stable?
It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in the Earth's atmosphere, at 0.934% (9340 ppmv). The complete octet (eight electrons) in the outer atomic shell makes argon stable and resistant to bonding with other elements.Which is a non metal that becomes isoelectronic with argon when it forms its most common ion?
Sulfur has 16 electrons. The nearest noble gas to sulfur is argon, which has an electron configuration of: 1s22s22p63s23p6. To be isoelectronic with argon, which has 18 electrons, sulfur must gain two electrons. Therefore sulfur will form a 2- ion, becoming S2-.What is isoelectronic with argon and neon?
Explanation: The Cl- ion has 18 electrons and is isoelectronic with argon while the F- ion has 10 electrons making it isoelectronic with neon.What are isoelectronic ions give examples?
Isoelectronic Definition Isoelectronic chemical species typically display similar chemical properties. Atoms or ions with the same electronic configurations are said to be isoelectronic to each other or to have the same isoelectronicity. Examples of Isoelectronic ions are:- Na+ and Mg+2, Ca+2 and Sc+3.