Where is water on the pH scale?
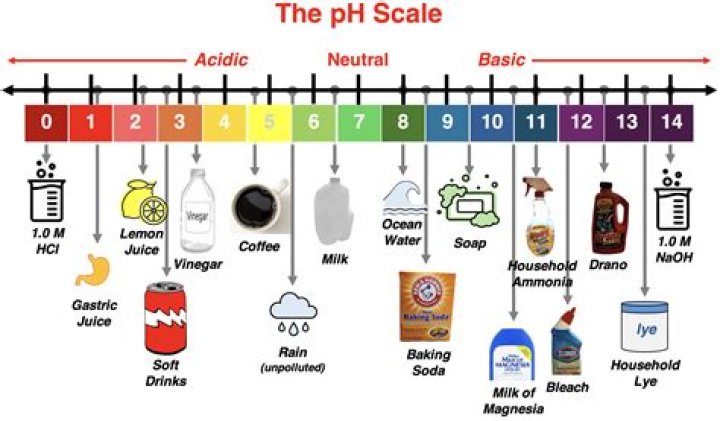
.
Just so, what is the pH of water?
6.5 to 8.5
Also Know, what causes pH in water? Carbon Dioxide and pH The degree of change depends on the alkalinity of thewater. Carbon dioxide is the most common cause ofacidity in water ¹5. Photosynthesis, respiration anddecomposition all contribute to pH fluctuations due to theirinfluences on CO2 levels.
In respect to this, what is the best pH level for drinking water?
Why 6 – 8.5 pH is ideal for DrinkingWater Water with a pH level between 6 and 8.5is safe to drink because it is neither acidic nor alkalineenough to be dangerous in the human body. Water with apH of less than 6 can be corrosive and filled with toxicmetals.
What is the full form of pH?
PH stands for Potential of Hydrogen. It refers tothe hydrogen ion concentration in a solution. It is the measure ofthe acidity or alkalinity of a solution. The PH value rangesfrom 0 to 14 on a pH scale.
Related Question AnswersWhat is the full meaning of pH?
A measure of acidity or alkalinity of water solublesubstances (pH stands for 'potential of Hydrogen'). ApH value is a number from 1 to 14, with 7 as the middle(neutral) point. Values below 7 indicate acidity which increases asthe number decreases, 1 being the most acidic.Does pH water matter?
The pH of your water should onlymatter if it's caustic enough to hurt you. Most commercialuncontaminated bottled water won't make you healthier orsick due to its pH. Depending on the source and processing,most bottled water stays between a pH of 5 to8.What is the importance of pH?
pH is an important quantity that reflectsthe chemical conditions of a solution. The pH can controlthe availability of nutrients, biological functions, microbialactivity, and the behavior of chemicals.Why is pH important to life?
pH is important because substances such asour stomach acids tend to be at a certain pH in order towork properly. pH is also important because it mustbe at certain levels in order for living organisms tosurvive.What pH water is good for?
Alkaline water has a higher pH level thanregular drinking water. Because of this, some advocates ofalkaline water believe it can neutralize the acid in yourbody. Normal drinking water generally has a neutralpH of 7. Alkaline water must also containalkaline minerals and negative oxidation reduction potential(ORP).What is the pH of milk?
6.5 to 6.7What pH is vinegar?
Vinegar is acidic. Vinegar's pH levelvaries based upon the type of vinegar it is. White distilledvinegar, the kind best suited for household cleaning,typically has a pH of around 2.5.Is lemon water alkaline?
Some sources say that lemon water has analkalizing effect, meaning that it can neutralize stomach acid,which may reduce acid reflux. However, this is not backed up byresearch. Lemon juice is acidic, with a pH of 3, whilewater has a pH of around 7, which is neutral. This means itis neither acidic nor alkaline.What is the pH of urine?
4.5 to 8.0What brand of water has the best pH?
- Aquafina: pH level 6.0. Aquafina water comes from municipalwater sources.
- Arrowhead Spring Water: pH level 6.8.
- Crystal Geyser: pH level 6.9.
- Dasani: pH level 5.6.
- Deer Park: pH level 5.5.
- Evian: pH level 7.4.
- Fiji: pH level 7.8.
- Nestle Pure Life: pH level 7.3.
Which foods are alkaline?
What is the alkaline diet?- Acidic: meat, poultry, fish, dairy, eggs, grains, alcohol.
- Neutral: natural fats, starches, and sugars.
- Alkaline: fruits, nuts, legumes, and vegetables.



