Where does the protease work?
.
Also, where does protease work in the body?
Proteases are released by the pancreas into the proximal small intestine, where they mix with proteins already denatured by gastric secretions and break them down into amino acids, the building blocks of protein, which will eventually be absorbed and used throughout the body.
Secondly, how are proteases activated in the stomach? When the protein material is passed to the small intestine, proteins, which are only partially digested in the stomach, are further attacked by proteolytic enzymes secreted by the pancreas. These enzymes are liberated in the small intestine from inactive precursors produced by the acinar cells in the pancreas.
Beside above, how does a protease work?
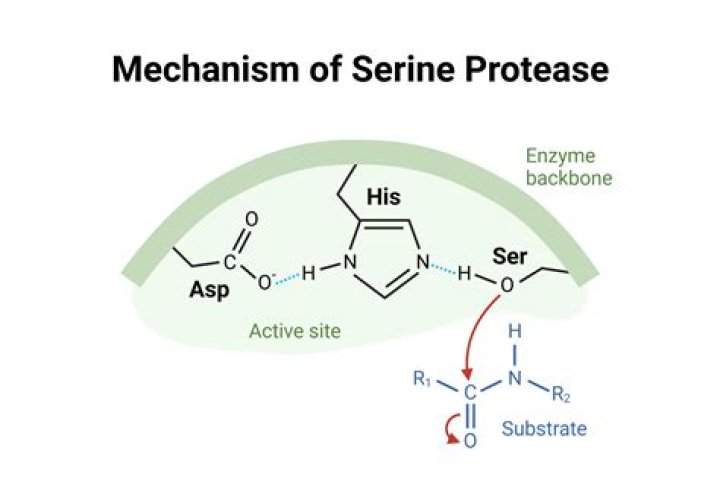
A protease (also called a peptidase or proteinase) is an enzyme that catalyzes (increases the rate of) proteolysis, the breakdown of proteins into smaller polypeptides or single amino acids. They do this by cleaving the peptide bonds within proteins by hydrolysis, a reaction where water breaks bonds.
What is the source of protease?
Two of the best food sources of proteolytic enzymes are papaya and pineapple. Papayas contain an enzyme called papain, also known as papaya proteinase I.
Related Question AnswersHow much protease should I take?
The recommended type and dosage of pancreatic enzymes must be individualized for each person. Most people should start off by taking 10,000 – 20,000 lipase units with snacks and 20,000 – 40,000 lipase units with meals.What are the symptoms of enzyme deficiency?
What are the symptoms of EPI ?- Diarrhea. EPI can cause problems with undigested food moving too quickly through the digestive tract.
- Gas and bloating.
- Stomach pain.
- Foul-smelling, greasy stools (steatorrhea)
- Weight loss.
How long does it take digestive enzymes to work?
The great thing about digestive enzymes is all they need to start working is food. They'll start to break down food molecules as soon as they come into contact with them. You should start to notice benefits within a few days! And remember: Your enzymes are only as good as what you feed them.Why do we need protease?
The Role of Protease Yes, protease helps break down protein in food into amino acids, which the body can then use for energy, but where proteases stand apart is the fact that they also play a number of other roles in essential processes, such as: Blood clotting. Recycling of proteins. Immune support.Can you take too many enzymes?
What happens if I take too many enzymes? Any enzymes that your body doesn't need will pass through you. If you think you've taken too many, make sure you drink plenty of fluids, and take your enzyme supplements at your next meal as usual. If you feel unwell, speak to your doctor or nurse.How are enzymes produced?
Enzymes are made from amino acids, and they are proteins. When an enzyme is formed, it is made by stringing together between 100 and 1,000 amino acids in a very specific and unique order. The chain of amino acids then folds into a unique shape. Other types of enzymes can put atoms and molecules together.Are digestive enzymes safe?
Many forms of digestive enzyme supplements are widely available over the counter, and they are mostly considered to be safe when taken as recommended. Some studies have suggested that bromelain, a digestive enzyme supplement made from pineapples, interferes with platelets in the bloodstream.What fruit contains protease enzymes?
Fruits like papaya, kiwifruit, pineapple and figs all contain enzymes called proteases. Proteases speed up the breakdown of proteins. Enzymes in papaya have a number of uses including tenderising meat and treating wounds.What is an example of protease?
Proteases are a protein-digestive enzyme that cleaves protein through hydrolysis, the addition of water to the peptide bond. An example of a protein-digesting enzyme may be seen in the protease called pepsin. Pepsin is one of two components of gastric juice. Pepsin works by attacking the exposed peptide bonds.What enzymes reduce inflammation?
Proteolytic enzymes, such as bromelain, papain, pancreatin, trypsin, chymotrypsin, and rutin, are essential regulators and modulators of the inflammatory response. Among their important actions is a seven- to ten-fold increase in the “appetite” of macrophages and in the potency of natural killer (NK) cells.What conditions does protease work best in?
The effect of pH| Enzyme | Optimum pH |
|---|---|
| Salivary amylase | 6.8 |
| Stomach protease (pepsin) | 1.5 - 2.0 |
| Pancreatic protease (trypsin) | 7.5 - 8.0 |