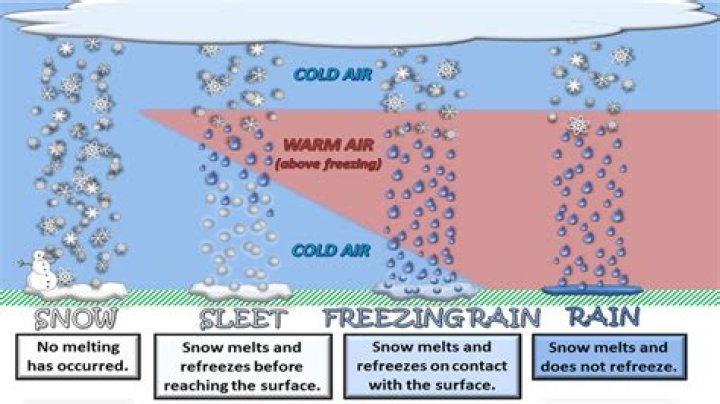
It is true, however, that most heavy snowfalls occur when there is relatively warm air near the ground—typically -9 degrees Celsius (15 degrees Fahrenheit) or warmer—since warmer air can hold more water vapor. Because snow formation requires moisture, very cold but very dry areas may rarely receive snow..
Similarly, when steam condenses the surrounding air is?
The energy released when gaseous water vapor condenses to form liquid water droplets is called latent heat. Latent heat from condensation causes an increase in air temperature surrounding the water droplets. The warmer air rises, causing the water vapor to condense when it meets cooler air at a higher altitude.
what cooks food faster in a pressure cooker quizlet? The higher temperature of boiling water under pressure cooks the food faster. The tight lid holds pressurized water vapor above the water surface, inhibiting boiling. Water must boil at a higher temperature, increased boiling point.
Consequently, during which phase changes is latent heat taken from the environment?
In the atmosphere, phase changes between liquid and gas are very important because of the large amount of latent heat involved. Latent heat released by water vapor during condensation in cloud formation is crucial for climate as it is part of the process that moves energy from the ground surface up into the atmosphere.
How does snow vary with temperature?
When water-vapor content of air remains constant, lowering air temperature causes an increase in relative humidity, and raising air temperature causes a decrease in relative humidity. The warm air rises above the cold air. How does snow vary with temperature? Cold snow is powdery and light, while warm snow is moist.
Related Question Answers
What is the amount of water vapor in the air?
The amount of water vapor in the air is called absolute humidity. The amount of water vapor in the air as compared with the amount of water that the air could hold is called relative humidity. This amount of space in air that can hold water changes depending on the temperature and pressure.What happens when air condenses?
Condensation is the process where water vapor becomes liquid. It is the reverse of evaporation, where liquid water becomes a vapor. Condensation happens one of two ways: Either the air is cooled to its dew point or it becomes so saturated with water vapor that it cannot hold any more water.What is after condensation?
This is followed by condensation, which is the process by which water vapor is changed back into liquid water. Then, water becomes precipitation, which is water falling from the clouds as rain, freezing rain, sleet, snow, or hail.What is the difference between water Vapour and steam?
Water vapor is little water droplets that exist in the air, while steam is water heated to the point that it turns into gas. In simplified science, both are referred to as the gaseous state of water. However, the main difference between them lies in temperature.What is the temperature of steam?
When water is heated at atmospheric pressure, its temperature rises until it reaches 212°F, the highest temperature at which water can exist at this pressure. Additional heat does not raise the temperature, but converts the water to steam.Are clouds water vapor?
Clouds are not water vapor. Water vapor is the gas state of H2O and is invisible. Clouds are collections of liquid water droplets or ice that are small enough to float. When the water vapor in the air gets cold enough, it condenses back into liquid in the forms of droplets.How do you test water vapor?
What are the Specific Tests for Water or Water Vapour? 1) Water or its vapour will turn cobalt chloride paper from blue to pink. anhydrous copper(II) sulfate crystals from white to blue. 3) Water melts at 0°C and boils at 100 °C.Is condensation pure water?
Water condensed from the air is almost totally pure. The only impurities are whatever dust is in the air around you and whatever it picks up from the surface it condenses on. Other than that, it's distilled water. But when it first condenses, it's totally safe.What is an example of latent heat?
Simple examples of latent heat: When we boil the water and the temperature remains at 100°C until the last drop evaporates, due to the latent heat of vaporization, the added heat in the water is absorbed and carried away by releasing vapor molecules. Latent heat of fusion of water: That is 334kJ/kg.How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.What does latent heat cause?
Latent heat. All pure substances in nature are able to change their state. Solids can become liquids (ice to water) and liquids can become gases (water to vapor) but changes such as these require the addition or removal of heat. The heat that causes these changes is called latent heat.How do you calculate phase change?
If the phase change is between solid and liquid the formula looks like q= mΔH fus and ΔH fus is called the heat of fusion. If the phase change is between liquid and gas the formula looks like q=mΔ vap and ΔH vap is called the heat of vaporization.What is the latent heat of water?
Latent heat of water is the energy required to change its state, also called its phase, without changing its temperature. The latent heat of fusion is the energy required to change a defined quantity of ice at 0 deg C from a solid to a liquid at the same temperature.Is freezing endothermic or exothermic?
Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and deposition are exothermic processes. Changes of state are examples of phase changes, or phase transitions. All phase changes are accompanied by changes in the energy of a system.What causes phase change water?
A phase change is occuring; the liquid water is changing to gaseous water, or steam. On a molecular level, the intermolecular forces between the water molecules are decreasing. The heat is providing enough energy for the water molecules to overcome these attractive forces.What is specific latent heat?
Specific Latent Heat. It takes a certain amount of energy to change the state of 1kg of water from solid to liquid. This amount of energy is called the Specific Latent Heat, lf, of water. The definition: "The amount of energy per kg (unit mass) required to change ice to water without change in temperature."Which phase change releases the most energy?
Latent Heat is the term describing the energy you get from exothermic phase changes. There are two phase changes where this heat energy is released: Condensation: When gas condenses to liquid the quantity of energy converted from chemical to heat is called the Heat of Vaporization or Δ Hv.What happens when you heat water at 100 C?
It increases the amount of internal energy because it does not allow the heat to boil the water. What happens when you heat water that is at 100∘C? The temperature stays constant until all the water is boiled. Liquid evaporates inside the refrigerator, absorbing energy.Is sublimation a cooling process?
Is it a heating or cooling process? Sublimation is when the fastest molecules at the surface of a solid have escape velocity and leave to become a gas. It leaves behind the slower, cooler molecules.