covalent bonding
.
Also to know is, what type of bond is present in so2?
SO2 is a covalent compund ! Covalent compounds are formed when atoms share electrons to complete their octet. Now, when two atoms that have similar electronegativities bond, the small difference between their respective electronegativities leads to a sharing of the bondingelectrons.
One may also ask, does sulfur dioxide have covalent bonds? Covalent compounds are formed when atoms share electrons to complete their octet. Sulfur shares a total of four valence electrons with the two oxygen atoms. Each oxygen atom shares two valence electrons with sulfur so sulfur dioxide is a covelent compound.
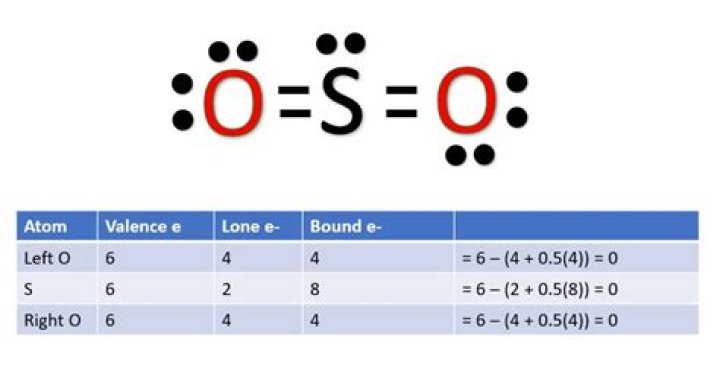
Additionally, does so2 have a coordinate bond?
It is best to say that SO2 has covalent bonds from the sulfur atom to each oxygen. The second O atom now forms an electron-pair bond with 2 electrons from the S & none from the O -- this shared electron pair bond is sometimes called a "coordinate covalent" bond -- and now the last O atom has a net closed-shell octet.
Is sulfur dioxide molecular or ionic?
Sulfur dioxide, SO2, is a molecular compound that contributes to acid rain, and CaCO3 is an ionic compound that can neutralize acid rain.
Related Question Answers
Is so2 bent or linear?
Carbon dioxide is linear, while sulphur dioxide is bent (V-shaped). In sulphur dioxide, as well as the two double bonds, there is also a lone pair on the sulphur. To minimise repulsions, the double bonds and the lone pair get as far apart as possible, and so the molecule is bent.Is so2 a polar molecule?
This means that there is one side (top or bottom) of the molecule that has both oxygen atoms on it, which gives it a slightly negative charge while the portion of the molecule that has the sulfur atom has a slightly positive charge. As a result of this, SO2 is polar.What type of bond is NaCl?
Ionic compounds
Is HCl ionic or covalent?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.What kind of bond is CO?
Carbon monoxide consists of one carbon atom and one oxygen atom, connected by a triple bond that consists of twocovalent bonds as well as one dative covalent bond. It is the simplest oxocarbon and is isoelectronic with the cyanide anion, the nitrosonium cation and molecular nitrogen.Is bcl3 polar or nonpolar?
The molecular geometry of BCl3 is trigonal planar with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Why is so2 bent and not linear?
CO2 is linear and the SO2 structure is bent, because CO2 has a negative oxygen on each side of the positive carbon they cancel each other out. In the SO2 structure the oxygen are not in lined with each other which means there a positive and negative end.How do I bond so2?
In the outer level, Sulphur has six electrons, and the Oxygen has four of them among which one electron is used for each bond. So total number of ten electrons in five pairs. To make bonds, four pairs are needed, so one pair remains alone. The two double bonds use two pairs each and form as a single unit.Is no2 linear?
Just like carbon dioxide? Except that NO2 is a V-shaped molecule, and CO2 is linear. two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.Is so2 trigonal planar?
For example, sulfur dioxide, SO2, electron-domain geometry is trigonal planar. This is because it has 3 electron domains - the 6 valence electrons for sulfur form 2 single bonds with 2 oxygen atoms and sulfur has one non-bonding lone pair. Since the difference in polarity is greater than zero, SO2 is a polar molecule.Is CCl4 polar or nonpolar?
CCl4 is an example of a nonpolar molecule. The four bonds of carbon tetrachloride (CCl4) are polar, but the molecule isnonpolar because the bond polarity is canceled by the symmetric tetrahedral shape. When other atoms substitute for some of the Cl atoms, the symmetry is broken and the molecule becomes polar.Is Sulphur dioxide in food bad for you?
Answer • Sulfur dioxide is used as a preservative, especially in dried fruits, but also in grapes (both fresh and in winemaking). Sulfur dioxide is considered to be safe; however, some people are sensitive to it, and it may even cause asthma symptoms in those who are predisposed.Are co2 and so2 polar or nonpolar?
CO2 is nonpolar, whereas SO2 is polar. As we know the hybridization of CO2 is sp which is linear in shape. Both oxygen having more electronegativity than carbon, so the electrons of both C-O bond shift towards oxygen .How many bond pairs and lone pairs are in so2?
It will have 2 bond pairs and 1 lone pair. S will make a double covalent bond with one O and a dative bond with the other. 2 remaining electrons of S will make 1 lone pair.Does co2 have a double bond?
Double Bonds A Double bond is when two atoms share two pairs of electrons with each other. As you can see from the picture below, Carbon dioxide has a total of 1 Carbon atom and 2 Oxygen atoms. Each Oxygen atom has 6 valence electrons whereas the Carbon atom only has 4 valence electrons.Is Sulphur a covalent?
Silicon Giant covalent lattice Break strong covalent bonds. Sulfur (S8) Simple molecular (lattice/covalent) Break weak London forces between the molecules.Is sulfur dioxide ionic bond?
Type of Bonding: Sulfur Dioxide is a process of covalent bonding, since Sulfur and Oxygen are both non-metals. The Sulfur is in the center surrounded by 2 Oxygen atoms.