1 Answer. The kinetic energy is the lowest at the solid state..
In this regard, what is the lowest energy state of matter?
solid
Furthermore, in which state does water have the lowest kinetic energy? Within a given substance, such as water, particles in the solid state have the least amount of kinetic energy. This is because they only vibrate in place. Particles in the liquid state move faster than particles in the solid state.
Hereof, what state of matter has the lowest entropy?
Entropy can be related to each state of matter. Because the particles of solids are found in an organized manner with little movement, it can be said that solids have the lowest entropy out of the three states of matter.
Which state of matter has highest amount of energy?
gas
Related Question Answers
What are the 15 states of matter?
Currently voted the best answer. Less familiar phases include: quark-gluon plasma; Bose-Einstein condensates and fermionic condensates; quantum spin Hall state; degenerate matter; strange matter; superfluids and supersolids; and possibly string-net liquids.What are the 20 states of matter?
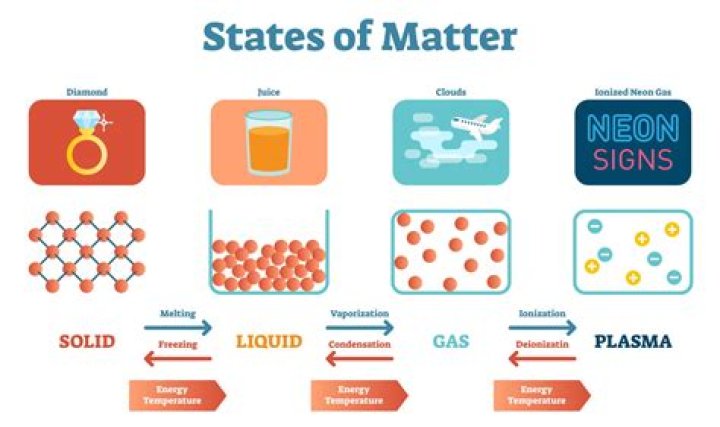
The classical states of matter are usually summarised as: solid, liquid, gas, and
plasma.
Low-energy states
- Solid: A solid holds a definite shape and volume without a container.
- Liquid: A mostly non-compressible fluid.
- Gas: A compressible fluid.
What are the 22 states of matter?
22 the states of matter - Chapter 8 The states of matter.
- What is matter?
- Examples for matter: ? 1)Solid ? 2)Liquid ? 3)Gas.
- A solid: ? Has definite mass.
- A liquid: ? Has a definite mass.
- A gas: ? Has a definite mass.
- ? Select one state of matter and imagine that it has been removed from the world.
Is fire a plasma?
Fire (flames) may contain plasma, albeit one that is a partially ionized plasma, and dominated by collisions: “Whether a plasma exists in a flame depends on the material being burned and the temperature”. fire) is shown as a plasma.Does air have mass?
Matter is anything that has mass and takes up space. So, in order to prove that air is matter, we need to prove that air has mass and takes up space. Although air has mass, a small volume of air, such as the air in the balloons, doesn't have too much. Air just isn't very dense.Is glass a solid or a liquid?
Glass, however, is actually neither a liquid—supercooled or otherwise—nor a solid. It is an amorphous solid—a state somewhere between those two states of matter. And yet glass's liquidlike properties are not enough to explain the thicker-bottomed windows, because glass atoms move too slowly for changes to be visible.What is it called when a solid changes to a liquid?
When a liquid changes into a solid, it is called solidification. When a gas changes into a solid, it is called deposition. When a liquid changes into gas, it is called evaporation.What is the 8th state of matter?
Bose - Einstein Condensate : A Bose–Einstein condensate (BEC) is a state of matter of a dilute gas of boson cooled to temperatures very close to absolute zero.What is the lowest entropy?
Solids have the fewest microstates and thus the lowest entropy. Liquids have more microstates (since the molecules can translate) and thus have a higher entropy.How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.What state has the highest entropy?
Explanation: Entropy by definition is the degree of randomness in a system. If we look at the three states of matter: Solid, Liquid and Gas, we can see that the gas particles move freely and therefore, the degree of randomness is the highest.Which substance has the highest entropy?
Answer and Explanation: Gases have the highest entropy. This is because gases can exist in a large number of different microstates.What phase has the highest enthalpy?
gas
Which gas has the highest entropy?
Greater the randomness of molecules of a substance greater is the entropy. Here hydrogen gas has more entropy as it shows more randomness/disorderliness due to less molar mass than all the given substances and also in the gas phase.What is the law of entropy tell us?
Entropy is one of the consequences of the second law of thermodynamics. The most popular concept related to entropy is the idea of disorder. Entropy is the measure of disorder: the higher the disorder, the higher the entropy of the system. This means that the entropy of the universe is constantly increasing.Which form of water has the lowest entropy?
The reason why ice forms is because the molecules in water are close enough together, either because of low temperature or very high pressure, that the lowest entropy state is a solid form. As you increase temperature at the right pressures, liquid is the lowest entropy state.How do I calculate entropy?
But entropy change is quoted in energy units of J. That means that if you are calculating entropy change, you must multiply the enthalpy change value by 1000. So if, say, you have an enthalpy change of -92.2 kJ mol-1, the value you must put into the equation is -92200 J mol-1.Which has least kinetic energy?
The kinetic energy is the lowest at the solid state since solid state exists at a lower temperature than liquid state and than gaseous state.What are 5 kinetic energy examples?
When you let go of that ball and let it fall, the potential energy converts into kinetic energy, or the energy associated with motion. There are five types of kinetic energy: radiant, thermal, sound, electrical and mechanical. Let's explore several kinetic energy examples to better illustrate these various forms.