What state is ammonium phosphate?
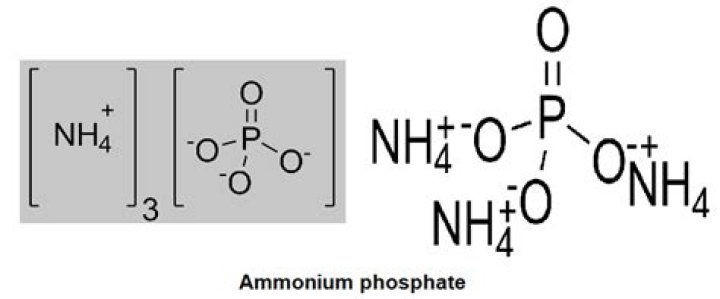
.
Considering this, is ammonium phosphate aqueous?
Ammonium phosphate is the salt of ammonia and phosphoric acid. It is soluble in water, and the aqueous solution on boiling loses ammonia and the acid phosphate (NH4)(H2PO4) is formed.
Subsequently, question is, how do you use ammonium phosphate? Ammonium phosphate is a fast release fertilizer and can be used when planting new grass, for maintenance, when overseeding or when renovating the lawn.
- Apply the ammonium phosphate when the ground is dry.
- Follow the instructions on the package to determine the rate in pounds per square footage that you will need.
Regarding this, is ammonium phosphate dangerous?
INHALATION: if exposed to ammonia fumes from diammonium phosphate, give artificial respiration and oxygen if needed; enforce rest. EYES: flush with water for at least 15 min.; if irritation persists, get medical attention.
How is ammonium phosphate produced?
A process for producing ammonium phosphates, comprising reacting by-product ammonia with phosphoric acid in solution under conditions to maintain in said solution a hydrogen ion concentration between the limits of pH=6.8 and pH=7.6, removing suspended solids from said solution, and then adding phosphoric acid to said
Related Question AnswersDoes ammonium phosphate dissolve in water?
It is soluble in water, and the aqueous solution on boiling loses ammonia and the acid phosphate (NH4)(H2PO4) is formed. Ammonium phosphate is used as an ingredient in some fertilizers as a high source of elemental nitrogen.Is ammonium phosphate an acid or base?
Ammonium phosphate, dibasic is an instance of the compound where there are two replaceable hydrogen atoms, whereas the monobasic example has only one hydrogen atom to donate to the base within the acid-base reaction of the ammonium and phosphate.What is magnesium phosphate used for?
The most common use for Magnesium Phosphate is to support the relaxation of the muscles and prevent cramping.Is ammonium phosphate soluble or insoluble in water?
H3PO4 + 3 NH3 → (NH4)3PO. (NH4)3PO4 is a colorless, crystalline solid. The solid, which has the odor of ammonia, is readily soluble in water. The salt converts to diammonium hydrogen phosphate (NH4)2HPO4.What is the pH of ammonium phosphate?
Properties| grade | ACS reagent |
|---|---|
| assay | ≥98.0% |
| impurities | ≤0.005% Ammonium hydroxide precipitate |
| ≤0.005% Insoluble matter | |
| pH | 7.7-8.1 (25 °C, 5% in solution) |
What is the charge of ammonia?
+1Is ammonium a metal?
Ammonium metal ions in a sea of electrons) at very high pressures, such as inside gas giant planets such as Uranus and Neptune. Under normal conditions, ammonium does not exist as a pure metal, but does as an amalgam (alloy with mercury).What is ammonium phosphate used for in food?
FOODS, FERTILIZER AND FIRES It's even listed as an ingredient in many commercially baked products. Not only can ammonium phosphate help feed humans, but it's used to feed plants as well. For years, it has been instituted as an effective fertilizer, with good water solubility and rapid dissolving abilities.What does ammonium phosphate do?
It is soluble in water, and the aqueous solution on boiling loses ammonia and the acid phosphate (NH4)(H2PO4) is formed. Ammonium phosphate is used as an ingredient in some fertilizers as a high source of elemental nitrogen. It is also used as a flame retardant in thermoplastic compositions.Is ammonium phosphate acidic?
Ammonium phosphate is an ammonium salt of orthophosphoric acid. It is a highly unstable compound with the formula (NH4)3PO4. Because of its instability, it is elusive and of no commercial value.Is ammonium phosphate a fertilizer?
Ammonium phosphate (16-20-0) adds nitrogen and phosphates to lawns that are lacking in those nutrients. Ammonium phosphate is a fast release fertilizer and can be used when planting new grass, for maintenance, when overseeding or when renovating the lawn.Is ammonium phosphate flammable?
Ammonium phosphate is an ammonium salt of orthophosphoric acid. It is a highly unstable compound with the formula (NH4)3PO4.Ammonium phosphate.
| Names | |
|---|---|
| GHS hazard statements | H302 , H319 |
| GHS precautionary statements | P264 , P270 , P280 , P301+312 , P305+351+338 , P330 , P337+313 , P501 |
| NFPA 704 (fire diamond) | 0 2 0 |
| Thermochemistry | |