What reacts with kmno4?
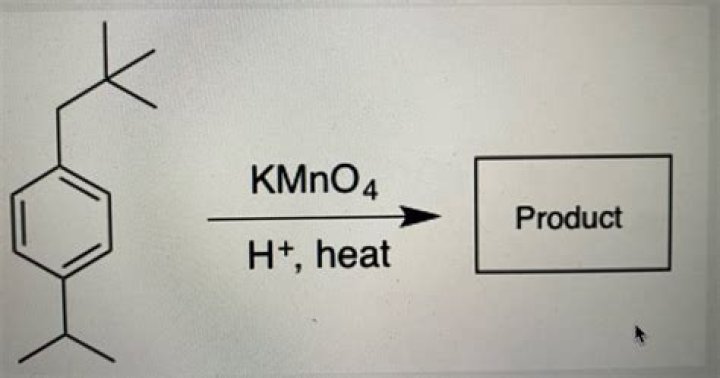
.
Furthermore, how does KMnO4 react with an alkene?
When a purple solution of the oxidizing agent KMnO4 is added to an alkene, the alkene is oxidized to a diol and the KMnO4 is converted to brown MnO2. The diol produced has two adjacent alcohol groups. Alkanes and aromatic compounds do not react with potassium permanganate.
One may also ask, what happens when ethene reacts with KMnO4? Alkenes react with potassium manganate(VII) solution in the cold. The colour change depends on whether the potassium manganate(VII) is used under acidic or alkaline conditions. If the potassium manganate(VII) solution is acidified with dilute sulphuric acid, the purple solution becomes colourless.
Also know, does ketone react with KMnO4?
KMnO4 also oxidizes phenol to para-benzoquinone. Exhaustive oxidation of organic molecules by KMnO4 will proceed until the formation of carboxylic acids. Therefore, alcohols will be oxidized to carbonyls (aldehydes and ketones), and aldehydes (and some ketones, as in (3) above) will be oxidized to carboxylic acids.
Does cyclohexene react with KMnO4?
The mechanisms in acidic and basic solutions appear to be quite different. A mechanism for basic solutions is suggested. Aqueous potassium permanganate reacts with cyclohexene to form a variety of products including cis-l,2-cyclohexane- diol.
Related Question AnswersWhat does KMnO4 and NaOH do?
Also note that osmium tetroxide (OsO4) does exactly the same reaction. The purpose of the NaOH is to assist in breaking up the intermediate manganate ester that forms after dihydroxylation. If this manganate ester is allowed to sit around, oxidative cleavage of the diol may occur.Does KMnO4 react with alkanes?
When a purple solution of the oxidizing agent KMnO4 is added to an alkene, the alkene is oxidized to a diol and the KMnO4 is converted to brown MnO2. Alkanes and aromatic compounds do not react with potassium permanganate.Can K2Cr2O7 oxidise alkene?
Take note that K2Cr2O7 cannot oxidise Alkenes at all this is because Pottasium Dichromate (K2Cr2O7) is not strong enough to oxidise Alkenes.Is KMnO4 a reducing agent?
No. In the MnO4- ion, the Mn has an oxidation state of +7. For it to act as a reducing agent, it needs to lose electrons to make another species gain them (so the oxidation state will at least move up to +8, OIL RIG=Oxidation is Loss, Reduction Is Gain).What is the role of KMnO4?
Potassium permanganate is used extensively in the water treatment industry. It is used as a regeneration chemical to remove iron and hydrogen sulfide (rotten egg smell) from well water via a "Manganese Greensand" Filter. "Pot-Perm" is also obtainable at pool supply stores and is used additionally to treat waste water.Why does alkene Decolourise KMnO4?
The acidified potassium manganate(VII) solution oxidises the alkene by breaking the carbon-carbon double bond and replacing it with two carbon-oxygen double bonds. The products are known as carbonyl compounds because they contain the carbonyl group, C=O.Why alkane does not react with KMnO4?
Alkanes, because of the presence of only sigma bonds and the absence of pi bonds, are quite stable and hence do not react with KMnO4. Alkenes are readily oxidised by cold dilute neutral or alkaline KMnO4 solution to give vicinal or 1,2-glycols while KMnO4 is itself reduced to MnO2.Why KMnO4 is used in acidic medium?
Thus, you can see that oxidizing effect of KMnO4 is maximum in acidic medium and least in basic medium as in acidic medium the reduction in oxidation state of Mn is max while it is the least in basic medium. Hence, acidic medium is used when you want strong oxidization and basic when very mild oxidization is required.Why KMnO4 is a self indicator?
So once all the permanganate ions are used up in the reaction, the solution loses its pink colour. This indicates the end of the reaction and hence potassium permanganate is called a self indicator as it acts as an indicator apart from being one of the reactants.Why KMnO4 is Coloured?
Potassium permanganate(KMnO4) is colored because it absorbs light in the visible range of the electromagnetic spectrum. This charge transfer takes place when a photon of light is absorbed, which leads to the purple color of the compound.Why is KMnO4 oxidizing agent?
KMnO4 is a strong oxidizing agent which can donate 4 oxygen atoms in acid media to achieve it's natural +2 oxidation state if a reducing agent (entity looking for electrons) is present. We obtain more electrons from permanganate ion (MnO4-) in acidic solution as compared to neutral and basic solution.Is KMnO4 an acid or base?
Potassium permanganate is neutral because it is formed by potassium hydroxide, which is a strong base and, permanganic acid, HMnO4, which I would like to stress is a STRONG acid. In water, the permanganate dissociates into K+ and MnO4- ions, in turn forming KOH and HMnO4, giving a neutral solution.Why ketone does not react with KMnO4?
Oxidation of Aldehydes. The presence of that hydrogen atom makes aldehydes very easy to oxidize. Because ketones do not have that particular hydrogen atom, they are resistant to oxidation, and only very strong oxidizing agents like potassium manganate(VII) solution (potassium permanganate solution) oxidize ketones.Do ketones react with Benedict's solution?
A few drops of the aldehyde or ketone are added to the reagent, and the mixture is warmed gently in a hot water bath for a few minutes.Using Fehling's solution or Benedict's solution.
| ketone | No change in the blue solution. |
|---|---|
| aldehyde | The blue solution produces a dark red precipitate of copper(I) oxide. |



