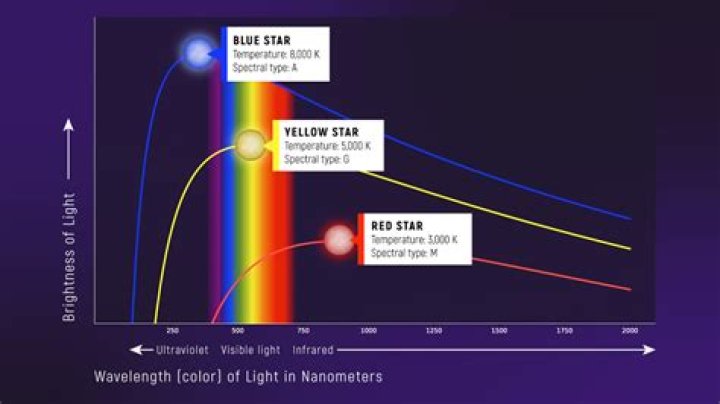
The spectrum of a star is composed mainly of thermal radiation that produces a continuous spectrum. The star emits light over the entire electromagnetic spectrum, from the gamma rays to radio waves. However, stars do not emit the same amount of energy at all wavelengths..
Keeping this in consideration, what are the 3 types of spectra?
The three main types of spectra: continuous, absorption, emission.
Additionally, what kind of spectra would be seen from an excited cloud of gas? Continuous Spectrum- A solid, liquid, or dense gas excited to emit light will radiate at all wavelengths and thus produce a continuous spectrum. A low-density gas excited to emit light will do so at specific wavelengths and this produces an emission spectrum.
Then, how do stars generate these types of spectra?
An absorption spectrum is produced when a continuum passes through "cooler" gas. Photons of the appropriate energies are absorbed by the atoms in the gas. The atmospheres of stars act as a cooler blanket around the hotter interior of a star so that typical stellar spectra are absorption spectra.
What type of spectrum is sunlight?
Such a spectrum from the Sun is known as the "visible spectrum", but it is just a small part of the light in the electromagnetic spectrum, which spans energies from radio waves to gamma-rays. The spectrum of the Sun appears as a continuous spectrum and is frequently represented as shown below.
Related Question Answers
Which color has the longest wavelength?
As the full spectrum of visible light travels through a prism, the wavelengths separate into the colors of the rainbow because each color is a different wavelength. Violet has the shortest wavelength, at around 380 nanometers, and red has the longest wavelength, at around 700 nanometers.How are spectra formed?
An emission spectrum is the line pattern formed when an element is excited and gives off energy. An absorption spectrum is formed when white light passes through a cool gas. The gas absorbs certain wavelengths of energy and allows others to pass through.What are the basic types of spectra?
There are three types of spectra which an object can emit: continuous, emission and absorption spectra. The examples of these types of spectra shown below are for visible light as it is spread out from purple to red, but the concept is the same for any region of the electromagnetic spectrum.What is the difference between spectra and spectrum?
A spectrum (plural spectra or spectrums) is a condition that is not limited to a specific set of values but can vary, without steps, across a continuum. The word was first used scientifically in optics to describe the rainbow of colors in visible light after passing through a prism.Why is sunlight a continuous spectrum?
So free electrons in the plasma medium in the sun's corona radiate Bremsstrahlung radiation due to statistical collisions and the output result is a continuous spectrum like back body radiation. This is also true for continuous radiation of hot objects and metals.Which spectrum is continuous?
A continuous spectrum contains many different colors, or wavelengths, with no gaps. Perfectly white light shined through a prism causes dispersion of the light, and we see a rainbow. This is a continuous spectrum.Why are line spectra unique to each element?
Each elements emission spectrum is distinct because each element has a different set of electron energy levels. The emission lines correspond to the differences between various pairs of the many energy levels. The lines (photons) are emitted as electrons fall from higher energy orbitals to lower energies.Why do stars die?
Stars die because they exhaust their nuclear fuel. The events at the end of a star's life depend on its mass. Really massive stars use up their hydrogen fuel quickly, but are hot enough to fuse heavier elements such as helium and carbon.What color are the hottest stars?
blue
How can you tell what a star is made of?
Each element absorbs light at specific wavelengths unique to that atom. When astronomers look at an object's spectrum, they can determine its composition based on these wavelengths. The most common method astronomers use to determine the composition of stars, planets, and other objects is spectroscopy.What can spectroscopy tell us about stars?
From spectral lines astronomers can determine not only the element, but the temperature and density of that element in the star. The spectral line also can tell us about any magnetic field of the star. The light from the stuff between the stars allows astronomers to study the interstellar medium (ISM).What causes the spectral lines of a star?
The presence of spectral lines is explained by quantum mechanics in terms of the energy levels of atoms, ions and molecules. Emission lines occur when the electrons of an excited atom, element or molecule move between energy levels, returning towards the ground state.What can we learn from stars?
(2) Another reason to study stars is that what we learn from other stars may help us understand our own Sun, which is also a star. (3) When we study stars, we also learn something about how they are born and die. This helps us understand how our own solar system was formed.How is spectroscopy used today?
Spectroscopy is used in physical and analytical chemistry because atoms and molecules have unique spectra. As a result, these spectra can be used to detect, identify and quantify information about the atoms and molecules. Spectroscopy is also used in astronomy and remote sensing on Earth.What do star spectra contain?
The spectrum of a star is composed mainly of thermal radiation that produces a continuous spectrum. The star emits light over the entire electromagnetic spectrum, from the gamma rays to radio waves. However, stars do not emit the same amount of energy at all wavelengths.How many spectrums of light are there?
Colors of the Visible Light Spectrum There are seven wavelength ranges in the visible spectrum that coordinate to a different color. Each visible color has a wavelength. As you move from red to violet, the wavelength decreases and energy increases. Here are the 7 from shortest to longest wavelength.How are absorption lines formed?
The intensity depends on the density and temperature of the gas. An absorption line is produced when a photon of just the right energy is absorbed by an atom, kicking an electron to a higher energy orbit. Other photons moving through the gas with the wrong energy will pass right on by the atoms in the thin gas.What do spectral lines represent?
A spectral line is a dark or bright line in an otherwise uniform and continuous spectrum, resulting from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. Spectral lines are often used to identify atoms and molecules.Why are the spectral lines different colors?
When its electron jumps from higher energy level to a lower one, it releases a photon. Those photons cause different colours of light of different wavelengths due to the different levels. Those photons appear as lines.