An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. A covalent bond involves a pair of electrons being shared between atoms..
Similarly, it is asked, what type of bond is a metal and a metal?
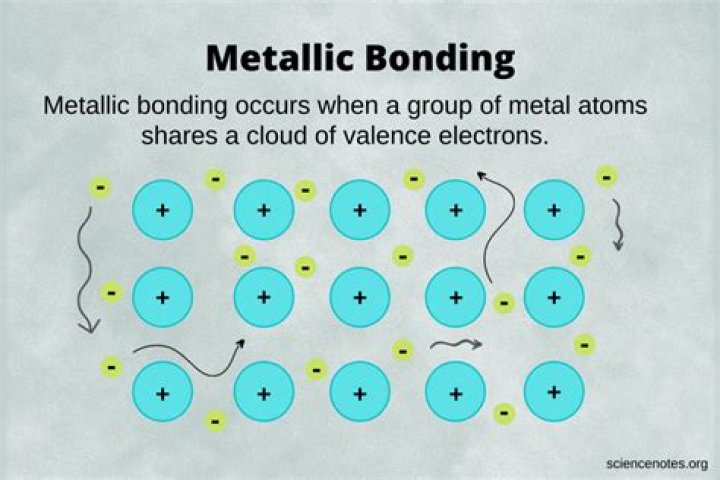
Pure metals or metal alloys form metallic bonds. A metallic bond is best described as positively charged nuclei engulfed in a sea of free electrons. These is why metals are good conductors, as all if the non-bonded atomic orbitals start to overlap and create an energy band known as the conduction band.
Secondly, what is an example of a metallic bond? The examples of metallic bond are iron, cobalt, calcium and magnesium, silver, gold, barium, platinum, chromium, copper, zinc, sodium, lithium and francium are some of the examples of metallic bonds. Covalent Bond. Metallic Bond. Chemical Reaction.
Similarly, you may ask, what kind of bond is 2 metals?
There are two types of atomic bonds - ionic bonds and covalent bonds. They differ in their structure and properties.
Covalent Bonds vs Ionic Bonds.
| Covalent Bonds | Ionic Bonds |
| Occurs between: | Two non-metals | One metal and one non-metal |
Can metals bond to other metals?
Metallic bonds occur among metal atoms. Whereas ionic bonds join metals to non-metals, metallic bonding joins a bulk of metal atoms. A sheet of aluminum foil and a copper wire are both places where you can see metallic bonding in action.
Related Question Answers
What type of bond is NaCl?
Ionic compounds
How is metallic bond formed?
Metallic bonds form between metal atoms due to the transfer of valence electrons (outer electrons) from one metal atom to the other- these commonly form regions of electron density in what are called s and p orbitals (don't worry about these, they're just interesting).What is metallic bonding simple?
A metallic bond is the sharing of many detached electrons between many positive ions, where the electrons act as a "glue" giving the substance a definite structure. It is unlike covalent or ionic bonding. The electrons and the positive ions in the metal have a strong attractive force between them.What is bond formation?
A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds.What holds metallic bonds together?
Metallic bond, force that holds atoms together in a metallic substance. The atoms that the electrons leave behind become positive ions, and the interaction between such ions and valence electrons gives rise to the cohesive or binding force that holds the metallic crystal together.How strong are metallic bonds?
Metals tend to have high melting points and boiling points suggesting strong bonds between the atoms. Even a metal like sodium (melting point 97.8°C) melts at a considerably higher temperature than the element (neon) which precedes it in the Periodic Table.What are some examples of ionic bonds?
Ionic bond examples include: - LiF - Lithium Fluoride.
- LiCl - Lithium Chloride.
- LiBr - Lithium Bromide.
- LiI - Lithium Iodide.
- NaF - Sodium Fluoride.
- NaCl - Sodium Chloride.
- NaBr - Sodium Bromide.
- NaI - Sodium Iodide.
Can two metals bond together?
Answer and Explanation: No. Two different elements, even if both are metals, cannot form a metallic bond together. They can only form mixtures of the two metals which we callCan 2 metals form a compound?
They don't really form compounds as compounds are formed when electrons are shared and metals do not share electrons with other metals. There is no point.How is a hydrogen bond formed?
A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. This makes hydrogen an electrically positive atom because it has a deficiency of electrons.What is meant by ionic bond?
Scientific definitions for ionic bond ionic bond. [ ī-ŏn′ĭk ] A chemical bond formed between two ions with opposite charges. Ionic bonds form when one atom gives up one or more electrons to another atom. These bonds can form between a pair of atoms or between molecules and are the type of bond found in salts.What is difference between ionic bond and covalent bond?
For stabilization, they share their electrons from outer molecular orbit with others. An ionic bond is formed between a metal and a non-metal. Covalent bonding is a form of chemical bonding between two non metallic atoms which is characterized by the sharing of pairs of electrons between atoms and other covalent bonds.Why is water polar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.What are the characteristics of metallic bonds?
Metallic bonding accounts for characteristics such as metals' strength, ductility, thermal and electrical resistivity, and conductivity, opacity and luster.What is a metallic bond and how is it formed?
Metallic Bonding In metallic bonds, the valence electrons from the s and p orbitals of the interacting metal atoms delocalize. That is to say, instead of orbiting their respective metal atoms, they form a “sea” of electrons that surrounds the positively charged atomic nuclei of the interacting metal ions.How is iron a metallic bond?
Iron is a metal and all metals have a type of bonding called metallic bonding which is different from covalent or ionic bonding. In metallic bonding the outer shells of adjacent atoms overlap, and the outer shell electrons are free to move through the lattice. The structure of iron is an example of a giant molecule.Is NaCl a metallic bond?
Ionic bonds occur when electrons are donated from one atom to another. Table salt (NaCl) is a common example of a compound with an ionic bond. You may also learn about a third type of bond. Metallic bonds occur between metal atoms.Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.Is steel a metallic bond?
Steel (at least mild steel) is composed of primarily iron so the metallic bonds are between iron atoms. Other more complex steel alloys like stainless steel include large amounts of nickel and chrome so the metallic bonds can be between iron and nickel, nickel and chrome, iron and chrome or nickel and chrome.