44.01 g/mol
.
Just so, how much does a liter of co2 weigh?
CO2 has a molecular weight of 44 g/mol 1 kg CO2 = 1000 g × (1 mol/44 g) = 22.7 mol CO2 V=nRT/P, V=(22.7)(0.0821)(300)/1 = 559 L CO2 at 27°C (300K), 1 atm This is a little more than half a cubic meter approximately equal to the volume of two bathtubs or the trunk of a large car.
Similarly, how much does co2 weigh in grams? You can view more details on each measurement unit: molecular weight of CO2 or grams This compound is also known as Carbon Dioxide. The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles CO2, or 44.0095 grams.
In this way, does carbon dioxide have weight?
Carbon dioxide has one carbon atom and two oxygen atoms, and a molecular weight of 44 grams per mole ( a certain number of molecules). The oxygen in the air is actually O2, or molecular oxygen, with a molecular weight of 32. Hence, carbon dioxide has a higher density, or is heavier than oxygen.
Is co2 heavier than air?
Carbon dioxide, also known by the chemical formula CO2, has a higher density than the other gases found in air, which makes CO2 heavier than the air. Air is composed of approximately 78 percent nitrogen, 21 percent oxygen, and less than 1 percent of other gases.
Related Question Answers
How big is a pound of co2?
One pound (454 grams) of carbon dioxide CO2 gas occupies 0.2426 m3, or 8.566 ft3, or 64 US gallons, or 243 liters – your choice of units! It would fill a cube 62.4 cm in all dimensions or a cube 24.6” across all dimensions.What is the volume of a ton of co2?
Volume calculation of one ton CO2. One ton of CO2 occupies 556.2m³ of volume. Every year the United States emits a 33.14cm high blanket of carbon dioxide over its land area. The combustion of each gallon of conventional gasoline produces 4.867m³ or 171.88ft3 of CO2.How do you calculate the weight of carbon dioxide?
Carbon Dioxide - Weight and Volume Equivalents
| Weight of Liquid or Gas | Volume of Gas at 70° (21°C) and 1 atm |
| lb | kg | cf |
| 1.000 | 0.454 | 8.743 |
| 2.205 | 1.000 | 19.278 |
| 2.238 | 1.015 | 19.566 |
Does air weigh anything?
It might not seem like it, but air has weight. Anything with mass has weight, and we know air has mass because (for example) we can feel it when the wind blows. The total weight of the atmosphere exerts a pressure of about 14.7 pounds per square inch at sea level.What is a volume of co2?
A volume is the space that the CO2 would take up at a pressure of one atmosphere (about 15 pounds per square inch) and at a temperature of 0° C (32° F) — that's Standard Temperature and Pressure (STP). An average carbonation level is 2.5 volumes of CO2. Beer holds more CO2 when it is cooler or under greater pressure.How many grams are a liter?
How many grams in 1 liter? The answer is 1000. We assume you are converting between gram [water] and liter. You can view more details on each measurement unit: grams or liter The SI derived unit for volume is the cubic meter.How much does a gallon of co2 weigh?
Therefore, to calculate the amount of CO2 produced from a gallon of gasoline, the weight of the carbon in the gasoline is multiplied by 44/12 or 3.7. Since gasoline is about 87% carbon and 13% hydrogen by weight, the carbon in a gallon of gasoline weighs 5.5 pounds (6.3 lbs. x .Do we breathe out carbon dioxide?
When we exhale, we breathe out less oxygen but more carbon dioxide than we inhale. The carbon we breathe out as carbon dioxide comes from the carbon in the food we eat. The carbon dioxide is dissolved in the blood, carried to the lungs by the circulation, and breathed out.Why is carbon dioxide bad for humans?
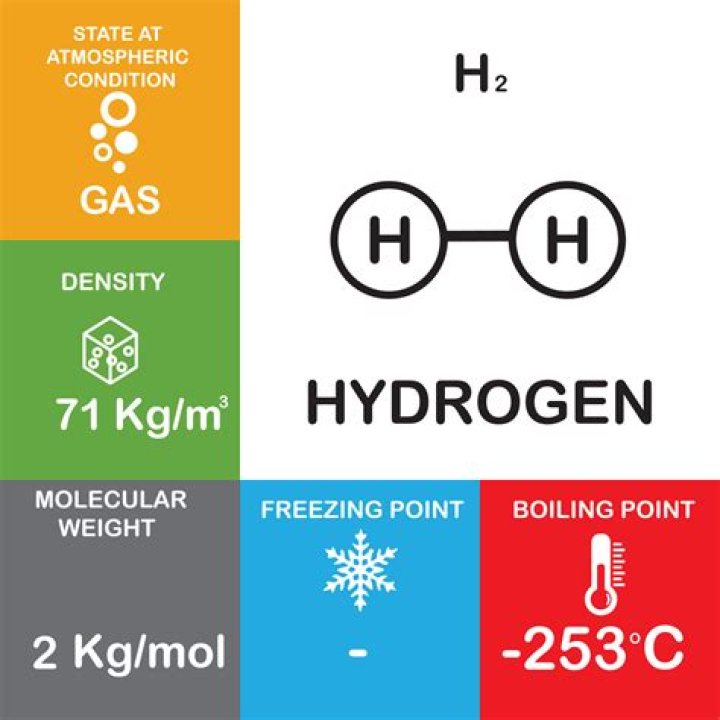
What are the potential health effects of carbon dioxide? Inhalation: Low concentrations are not harmful. Higher concentrations can affect respiratory function and cause excitation followed by depression of the central nervous system. A high concentration can displace oxygen in the air.Is hydrogen heavier than air?
Hydrogen has only about 7% of the mass of atmospheric air. making it much less dense than air. This is what made Hydrogen the choice of gas for the lighter than air ships. Now lighter than air ships use Helium with a mass of 4 grams./ 22.4 liters of volume.Is co2 flammable?
Carbon dioxide is a colorless, odorless, faintly acidic-tasting, non-flammable gas. It can be deadly even when normal oxygen levels are present. Even slow leaks can cause hazardous concentrations of carbon dioxide.Can we remove co2 from the atmosphere?
There are many forms of negative emissions, but most likely the only way to remove enough CO2 will be to pull it directly out of the air and bury it underground in saline aquifers, a process known as carbon capture and sequestration (CCS).How many pounds are in a ton of co2?
Gas doesn't weigh that much! So what do they mean by a “ton of carbon dioxide”? I figured since it was also sometimes spelled “tonne”, maybe it was an obscure European metric unit of volume. Amazingly, no, they actually mean a ton, as in 2000 pounds.What is molar mass of carbon?
12.0107 u
How much co2 does a ton of coal emit?
For example, coal with a carbon content of 78 percent and a heating value of 14,000 Btu per pound emits about 204.3 pounds of carbon dioxide per million Btu when completely burned. Complete combustion of 1 short ton (2,000 pounds) of this coal will generate about 5,720 pounds (2.86 short tons) of carbon dioxide.How are carbon emissions calculated?
Carbon dioxide emissions per therm are determined by converting million British thermal units (mmbtu) to therms, then multiplying the carbon coefficient times the fraction oxidized times the ratio of the molecular weight of carbon dioxide to carbon (44/12). 0.1 mmbtu equals one therm (EIA 2018).How do they measure co2 emissions?
The carbon dioxide equivalent (CO2e) allows the different greenhouse gases to be compared on a like-for-like basis relative to one unit of CO2. CO2e is calculated by multiplying the emissions of each of the six greenhouse gases by its 100 year global warming potential (GWP).What is the weight of carbon dioxide?
44.01 g/mol