What is the strongest bond in chemistry?
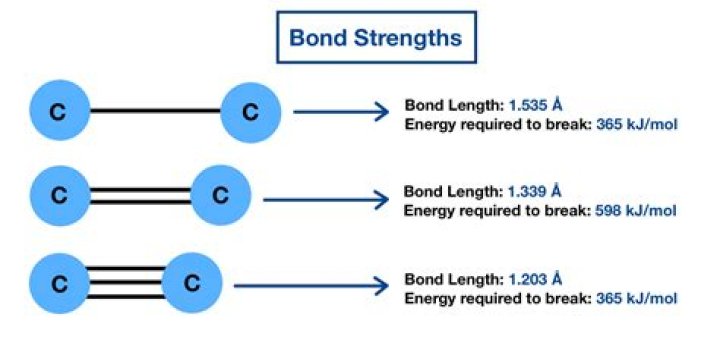
.
Consequently, which is stronger ionic or covalent bonds?
Covalent is stronger because the 2 atoms involve share 2 or more outer shell electrons. Covalent bonds hold all of your biomolecules together. Ionic bonds are formed when a valence outer shell electron is transferred from one atom to another - a much weaker interaction.
Similarly, what is the weakest bond in chemistry? The weakest of the intramolecular bonds or chemical bonds is the ionic bond. next the polar covalent bond and the strongest the non polar covalent bond.
Regarding this, what is the strongest bond in organic chemistry?
The high electronegativity of fluorine (4.0 for F vs. 2.5 for carbon) gives the carbon–fluorine bond a significant polarity/dipole moment. The bond is labeled as "the strongest in organic chemistry," because fluorine forms the strongest single bond to carbon.
Why are ionic bonds weak in water?
Ionic bonds within a salt crystal may be quite strong. However, if the crystal of salt is dissolved in water, each of the individual ions becomes surrounded by water molecules, which inhibit oppositely charged ions from approaching one another closely enough to form ionic bonds. In water, ionic bonds are very weak.
Related Question AnswersWhat are examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.
Are covalent bonds strong?
Covalent bonds between atoms are quite strong, but attractions between molecules/compounds, or intermolecular forces, can be relatively weak. Covalent compounds generally have low boiling and melting points, and are found in all three physical states at room temperature.Are covalent bonds strong or weak?
Covalent and ionic bonds are both typically considered strong bonds. However, other kinds of more temporary bonds can also form between atoms or molecules. Two types of weak bonds often seen in biology are hydrogen bonds and London dispersion forces.Are ionic bonds the strongest?
Ionic Bonding. They tend to be stronger than covalent bonds due to the coulombic attraction between ions of opposite charges. To maximize the attraction between those ions, ionic compounds form crystal lattices of alternating cations and anions.Why are covalent bonds strong?
An atom that shares one or more of its electrons will complete its outer shell. Covalent bonds are strong - a lot of energy is needed to break them. Both nuclei are strongly attracted to the shared pair of electrons in the covalent bond, so covalent bonds are very strong and require a lot of energy to break.Why are covalent bonds weak?
Covalent compounds are the ones having strong intra-molecular bonds. This is because the atoms within the covalent molecules are very tightly held together. Each molecule is indeed quite separate and the force of attraction between the individual molecules in a covalent compound tends to be weak.How do you identify a covalent bond and ionic bond?
There is a couple different ways to determine if a bond is ionic or covalent. By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.Which type of bond is strongest?
| Bond Strength | Description | |
|---|---|---|
| Covalent | Strong | Two atoms share electrons. |
| Ionic | Moderate | Oppositely charged ions are attracted to each other. |
| Hydrogen | Weak | Forms between oppositely charges portions of covalently bonded hydrogen atoms. |



