What is the size of atom and nucleus?
.
Keeping this in consideration, is an atom bigger than a nucleus?
An atomic nucleus is much, much smaller than an atom. The cloud of electrons that "orbit" the nucleus and define the "size" of an atom is roughly 100,000 times as large as that atom's nucleus!
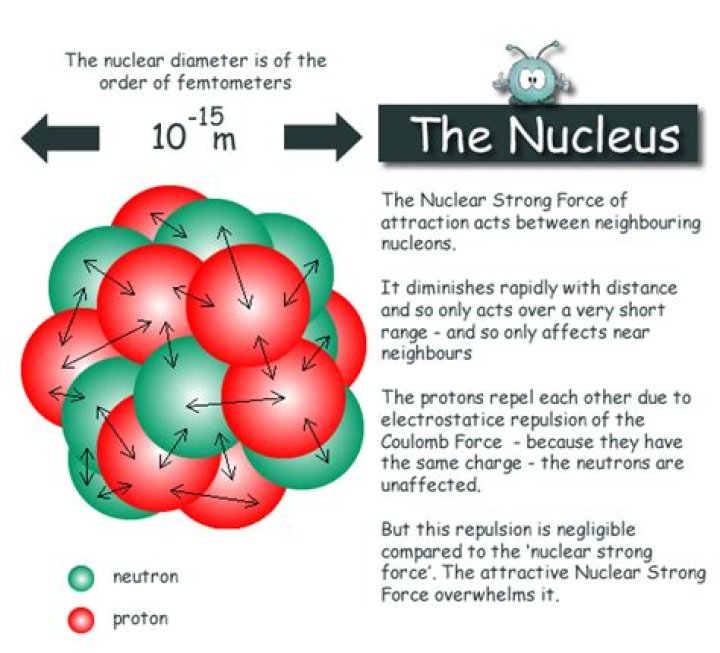
Also Know, what does a nucleus do in an atom? The Nucleus: The Center of an Atom. The nucleus, that dense central core of the atom, contains both protons and neutrons. Electrons are outside the nucleus in energy levels. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge.
Thereof, what is the size of an atom?
An atom is one of the basic units of matter. Everything around us is made up of atoms. An atom is a million times smaller than the thickest human hair. The diameter of an atom ranges from about 0.1 to 0.5 nanometers (1 × 10−10 m to 5 × 10−10 m).
What is meant by the nucleus of an atom?
In chemistry, a nucleus is the positively charged center of the atom consisting of protons and neutrons. It's also known as the "atomic nucleus". Nearly all the mass of an atom is contained within the nucleus since protons and neutrons have much more mass than electrons.
Related Question AnswersHow big is the largest atom?
The elements with the largest atomic radii are located towards the left and bottom of the periodic table (i.e. at the bottom of Group 1). Thus, in theory, the largest atom should be Francium.How big is an electron?
Fundamental properties The invariant mass of an electron is approximately 9.109×10−31 kilograms, or 5.489×10−4 atomic mass units.How many inches is an atom?
So or 10 billion hydrogen atoms would fit in a centimeter. Using 1 inch = 2.54 centimeters, we get that 25.4 billion hydrogen atoms would make 1 inch.What is atomic mass number?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number is different for each different isotope of a chemical element.How small is the nucleus of an atom?
The nucleus of an atom is about 10-15 m in size; this means it is about 10-5 (or 1/100,000) of the size of the whole atom. A good comparison of the nucleus to the atom is like a pea in the middle of a racetrack. (10-15 m is typical for the smaller nuclei; larger ones go up to about 10 times that.)Which is bigger atom or neutron?
Neutrons are uncharged particles found within all atomic nuclei (except for hydrogen). A neutron's mass is slightly larger than that of a proton. Like protons, neutrons are also made of quarks — one "up" quark (with a positive 2/3 charge) and two "down" quarks (each with a negative one-third charge).How small is an atom comparison?
The Tiny Size of Atoms The average diameter of an atom is 0.1 to 0.5 nanometers. One meter contains 1,000,000,000 nanometers. Smaller units, such as centimeters and millimeters, typically used to measure small objects that can fit within your hand, are still much larger than a nanometer.What are atoms made of?
An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud.How big is a hydrogen atom?
How big is a hydrogen atom? The radius of a hydrogen atom is known as the Bohr Radius, which is equal to . 529 × 10-10 meters. That means that a hydrogen atom has a volume of about 6.2 × 10-31 cubic meters.How many atoms are in a human?
In summary, for a typical human of 70 kg, there are almost 7*1027 atoms (that's a 7 followed by 27 zeros!) Another way of saying this is "seven billion billion billion." Of this, almost 2/3 is hydrogen, 1/4 is oxygen, and about 1/10 is carbon. These three atoms add up to 99% of the total!Can you see an atom?
Atoms are so small that we cannot see them with our eyes (i.e., microscopic). To give you a feel for some sizes, these are approximate diameters of various atoms and particles: atom = 1 x 10-10 meters. neutron or proton = 1 x 10-15 meters.Who discovered atom?
Democritus was a Greek philosopher who was the first person to use the term atom (atomos: meaning indivisible). He thought that if you take a piece of matter and divide it and continue to divide it you will eventually come to a point where you could not divide it any more.How big is a helium atom?
Labs use the Helium atom (Helium gas) for testing porosity and permeability of materials as it is the smallest of the single atom gases, with an atomic radius of 31pm, or diameter of 62pm.How small is a quark?
While the size of protons and neutrons is of the order of a Fermi (10−15 m), the size of quarks is ~10−18 m. It is deemed that quarks are composed of smaller particles – preons.What contains nucleus of an atom?
The nucleus, that dense central core of the atom, contains both protons and neutrons. Electrons are outside the nucleus in energy levels. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. A neutral atom contains equal numbers of protons and electrons.What are 4 types of atoms?
There are many different types of atoms, each with its own name, atomic mass and size. These different atoms are called chemical elements.Atom.
| Helium atom | |
|---|---|
| Components | Electrons and a compact nucleus of protons and neutrons |