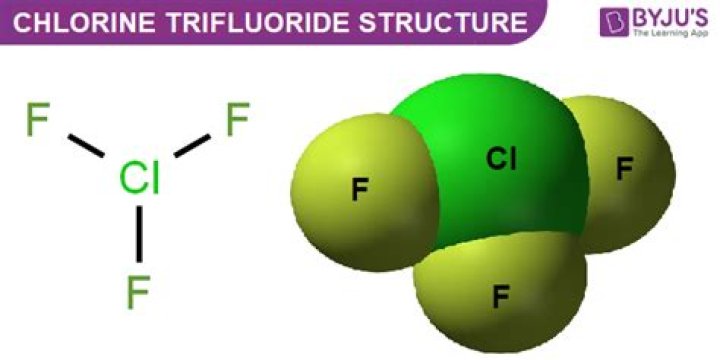
Chlorine trifluoride has 5 regions of electron density around the central chlorine atom (3 bonds and 2 lone pairs). These are arranged in a trigonal bipyramidal shape with a 175° F(axial)-Cl-F(axial) bond angle. The two lone pairs take equatorial positions because they demand more space than the bonds..
Keeping this in view, what is the structure of chlorine trifluoride?
ClF3
Similarly, what is the electron pair geometry for CL in ClF3? The electron geometry of chlorine trifluoride is trigonal bipyramidal with a 175° F-Cl-F bond angle.
Similarly, what does chlorine trifluoride look like?
Chlorine trifluoride appears as a colorless gas or green liquid with a pungent odor. Boils at 53°F. It reacts with water to form chlorine and hydrofluoric acid with release of heat.
What is chlorine trifluoride used for?
Uses: Chlorine trifluoride is use in electronic industry to clean semiconductors from chemical deposition. During the second world war it was studied as a chemical weapon. Most of chlorine trifluoride is used in nuclear fuel processing.
Related Question Answers
How is chlorine trifluoride stored?
The only known way to store chlorine trifluoride “safely”, which we use in the loosest possible sense, is to put it inside of a sealed containers made of steel, iron, nickel or copper which are able to contain the chemical safely if they're first treated with flourine gas.Is chlorine trifluoride polar or nonpolar?
The molecular geometry of ClF3 is T-shaped with asymmetric charge distribution around the central atom. Therefore ClF3 is polar.Is chlorine trifluoride illegal?
The History of Chlorine Trifluoride Though, back in 1930, scientists Otto Ruff and H. Krug isolated a compound which would prove to be more reactive than fluorine. Fortunately, since the war, the compound has been banned under the Chemical Weapons Convention.Is ClF3 trigonal planar?
In this case, the three bonds must be single covalent bonds and the atom orients itself to form what is called a trigonal planar or pyramidal geometry, where the atoms form a 'T' or pyramid respectively. But, to answer your question, t-shaped molecules are indeed planar. For example, chlorine triflouride or ClF3.Is h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.Is PCl3 planar?
No. PCl3 is not trigonal planar. It is trigonal pyramidal in shape. This is due to PCl3 being sp3 hybridized.Does ClF3 have a dipole moment?
the formula ClF3 . because, if the molecule had a trigonal planar structure, the molecule would be symmetric with dipoles that cancel out and produce a net dipole of zero (i.e., a nonpolar molecule), which is not consistent with the observation that the ClF3 molecule does have a dipole moment Page 5 .What is the name of cl2o7?
Dichlorine heptoxide is the chemical compound with the formula Cl2O7. This chlorine oxide is the anhydride of perchloric acid.Does chlorine explode?
Chlorine is not flammable, but it reacts explosively or forms explosive compounds with many common substances.Does fluorine explode?
Hydrogen and fluorine will explode without any contact and in complete darkness. Plenty of labs and chemical processing plants have been rocked by hydrogen and fluorine explosions. (In one case, fluorine started eating into its own containment canister, which created hydrogen gas, which exploded the canister.)How dangerous is fluorine?
If fluorine is absorbed too frequently, it can cause teeth decay, osteoporosis and harm to kidneys, bones, nerves and muscles. Fluorine gas is released in the industries. This gas is very dangerous, as it can cause death at very high concentrations. At low concentrations it causes eye and nose irritations.Why does clf3 have 2 lone pairs?
Chlorine trifluoride has 5 regions of electron density around the central chlorine atom (3 bonds and 2 lone pairs). These are arranged in a trigonal bipyramidal shape with a 175° F(axial)-Cl-F(axial) bond angle. The two lone pairs take equatorial positions because they demand more space than the bonds.Does clf3 follow octet rule?
why clf3 does not follow octet rule. The 'octet' rule is based upon available ns and np orbitals for valence electrons (2 electrons in the s orbitals, and 6 in the p orbitals). Hence, the third period elements occasionally exceed the octet rule by using their empty d orbitals to accommodate additional electrons.What chemical is flammable with water?
hydrogen gas
Does chlorine react with metal?
Chlorine reacts with most metals and forms metal chlorides, with most of these compounds being soluble in water. Examples of insoluble compounds include AgCl and PbCl2. Gaseous or liquid chlorine usually does not have an effect on metals such as iron, copper, platinum, silver, and steel at temperatures below 230°F.What is chlorine and fluorine?
A chlorine fluoride is an interhalogen compound containing only chlorine and fluorine.Is a chlorine spill dangerous?
Because of its widespread use in industrial and commercial locations, exposure to chlorine could occur from an accidental spill or release, or from a deliberate terrorist attack. The most harmful route of exposure is from breathing chlorine gas.Why is ClF polar?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). For these molecules, the applicable intermolecular interactions are dipole and induced dipole interactions.What is the hybridization of ClF?
The original ClF molecule is not hypervalent, and does have sp3 hybridization on both atoms. When chlorine turns into a cationic center, it is keeping its sp3 hybridization and is able to accept two fluorine atoms. Resonance allows the axial fluorine atoms to be kept in place by the chlorine atom in ClF3.