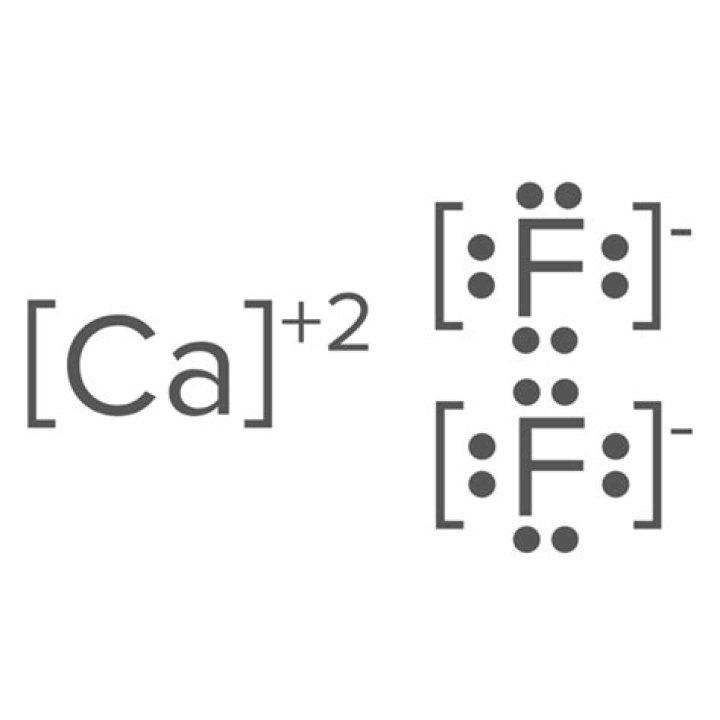Calcium fluoride is the inorganic compound of the elements calcium and fluorine with the formula CaF2..
Similarly, what is the chemical formula of fluorspar?
CaF2
Subsequently, question is, is CaF2 molecular or ionic? Calcium Fluoride. Calcium fluoride (CaF2) is an insoluble ionic compound composed of Ca2+ and F− ions. It occurs naturally as the mineral “Fluorite” (also called fluorspar) and as “Blue-John”. This salt is the source of most of the world's fluorine.
In respect to this, what is the structure of CaF2?
Calcium fluoride is an ionic crystal with the fluorine anions in a simple cubic array and calcium cations in half of the cubic sites of the structure. The calcium ions can also be thought of as being on an "expanded" fcc lattice with the fluorine ions causing their lattice separation to be 0.39 nm.
Is CaF2 polar or nonpolar?
In the Linear shape CaF2 is non-polar, but in the bent shape it becomes polar.
Related Question Answers
Where is fluorspar found?
Fluorite is found worldwide in China, South Africa, Mongolia, France, Russia, and the central North America. Here, noteworthy deposits occur in Mexico, Illinois, Missouri, Kentucky and Colorado in the United States.What is fluorspar for?
Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals.Is fluorite used in toothpaste?
The fluoride in toothpastes is a chemical made from the mineral fluorite. It is thought that fluoride reduces tooth decay, so if you clean your teeth every day, you shouldn't need fillings the next time you go to the dentist!Is fluorite safe to wear?
However, despite being a softer collector's stone, fluorite is still quite suitable for protected jewelry such as pendants, brooches or earrings. Multicolor banded beads of fluorite are sometimes used in bracelets, but care should always be taken when wearing fluorite.What is apatite used for?
Uses of Apatite as Phosphate Rock Most of the phosphate rock mined throughout the world is used to produce phosphate fertilizer. It is also used to produce animal feed supplements, phosphoric acid, elemental phosphorous, and phosphate compounds for the chemical industry.What minerals are in toothpaste?
Minerals can be found in your toothpaste, and they're there for good reason. Calcium and phosphorous (in the form of phosphate), along with fluoride, are included in toothpaste because they play a key role in keeping your teeth from becoming soft and yucky, which makes it easy for cavities to form..How fluorite is formed?
Fluorite crystals formed 150–200 million years ago when hot water containing fluorine and other minerals was forced up through cracks in the earth where it interacted with the calcium-rich limestone bedrock. Crystals formed along cracks and in other open spaces in the rock.What is apatite family?
Apatite family is a group that mainly contain phosphorus as a prime mineral. Along with phosphorus mainly elements are present along with it and hence the name. e.g. Flourapapite, hydroxylapatite, Chlorapatite etc. containing F- , OH- and Cl- respectively.What type of bond is CaF2?
ionic bond
What is CCP lattice?
CCP stands for cubic closed packing , FCC is for face centered cubic structure and HCP is for hexagonal cubic packing . Now , HCP and CCP are one of the forms in which a cubic lattice is arranged and FCC is one of the types of unit cells(in general)Why is CaF2 insoluble in water?
Why is CaCl2 more soluble in water than CaF2? A few water molecules do not have enough strength to get in between and dissolve it. Chloride is less electronegative, thus CaCl2 has weaker bonds, So weaker the bond strength, greater the solubility – at least that's the expected.Is BaF2 soluble?
What is the molar solubility of barium fluoride ( BaF2 ) in water? The solubility-product constant for BaF2 is 1.7 x 10^-6 at 25 C.Is CaF soluble?
Calcium Fluoride Goes Soluble. Fluorspar, CaF2, is the only raw-material source for industrial fluorine compounds.Is CaF2 acidic basic or neutral?
Ex: NaCl is a neutral salt because NaOH is a strong base and HCl is a strong acid. CaF2 is slightly basic. The reason is HF is a weak acid ( but it is very corrosive). When dissolved in water, the F- ion formed will undergoes hydrolysis reaction with water molecules forming HF and OH-.Is CaF2 aqueous?
Fluorite, or calcium fluoride (CaF2), another simple halide, is found in limestones that have been permeated by aqueous solutions containing the fluoride anion.What is the coordination number of CaF2?
eight
Is h2o covalent or ionic?
H2O, more commonly known as water, is a covalent compound. This type of compound is the result of atoms, usually from nonmetal elements, sharing electrons. Water has a special type of covalent bond called a polar covalent bond.What type of bond is KCl?
Chemical bonding is the process that links elements together to form compounds. KCl is an example of ionic bonding. Below is a simple diagram of potassium and chlorine atoms. Electrons are shown to be arranged in rings around a central nucleus.What type of bonding is NaOH?
ionic