What is the molecular geometry of so3 2?
.
Herein, what is the molecular geometry of so3?
Sulfur trioxide has trigonal planar molecular geometry since all three S=O. bonds are nearly double bonds and all are the same length, and energy, and are 120 degree apart. Using VSEPR theory we use the sigma bonds to establish the geometry: three bonding pairs and no lone pairs on the central atom = trigonal planar.
Similarly, is so32 tetrahedral? Explanation: For sulfur trioxide we must distribute 3×6=18⋅valence electrons . And thus O=..S(−OH)2 the central sulfur is sp3−hybridized , and the electron pairs assume a tetrahedral geometry. But molecular geometry is described in terms of ATOMS not electron pairsand so the geometry around sulfur is pyramidal
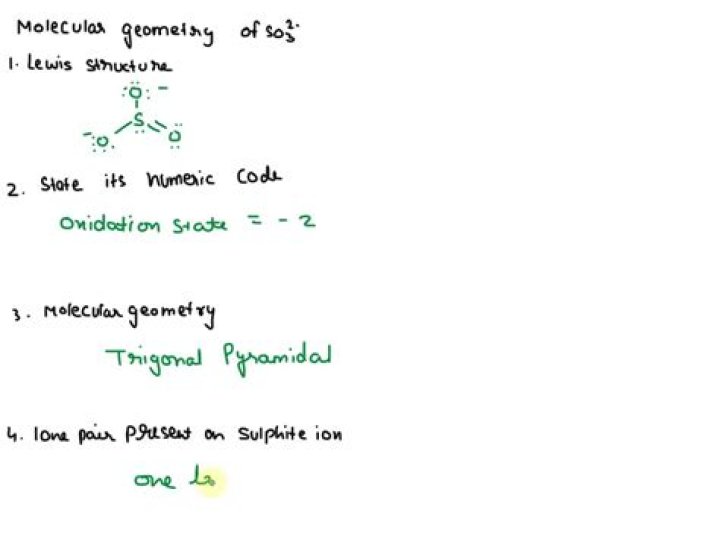
People also ask, what is the Lewis structure for so3 2?
The Lewis Dot Structure for SO3 2-: The sulfite anion (SO3 2-) is present in wines, and is used as preservative in certain foods. The accepted Lewis structure predicts both single and double bonds between S and O atoms involving available valence electrons.
What is the hybridization of so3 2?
It is sp3 , trigonal planar. Sulfur loves to break the octet rule. so be generous with double bonds. So3-2 is a different specie, it is called the sulfite ion. Here the hybridization is sp3d because the lone pair resides on the sulfur atom.
Related Question AnswersIs so3 2 trigonal planar?
Originally Answered: How do I show that SO3 2 is a trigonal pyramidal shape with the help of VSEPR theory? For SO3 2, the central atom is going to be the one that forms the most bonds; in this case, it is sulfur. Since sulfur is our central atom, the three oxygens are going to be bonded to sulfur.Why is so2 bent and not linear?
CO2 is linear and the SO2 structure is bent, because CO2 has a negative oxygen on each side of the positive carbon they cancel each other out. In the SO2 structure the oxygen are not in lined with each other which means there a positive and negative end.What is the bond order of so3 2?
SO3 bond order is 2 as covalent bond is replaced by double bond according to modern theory and if it comes in advance answer would be 2 untill it is mentioned to tell by LDS. according to LDS answer will be 4/3.What is the Lewis structure for ch2o?
Let's do the Lewis structure for CH2O, methanal or formaldehyde. Start with the valence electrons. Looking at the periodic table, Carbon has 4. Hydrogen, in group 1, has 1 and Oxygen, in group 6 or 16, has 6; but we have two Hydrogens, so let's multiply that by 2.What is the Lewis structure for clo4?
Drawing the Lewis Structure for ClO4- (Perchlorate Ion) A commonly used perchlorate is ammonium perchlorate (NH4ClO4) found in solid rocket fuel. There are 32 valence electrons available for the Lewis structure for ClO4-.Is so3 trigonal pyramidal?
The molecular geometry of SO3 is trigonal planar with symmetric charge distribution on the central atom. Therefore SO3 is nonpolar. Sulfur Trioxide on Wikipedia.What is the structure of bf3?
The geometry of molecule of BF3 is 'Trigonal Planar. ' With the reference of Chemistry, 'Trigonal Planar' is a model with three atoms around one atom in the middle. It's like peripheral atoms all in one plane, as all three of them are similar with the 120° bond angles on each that makes them an equilateral triangle.Is so3 a Lewis acid or base?
The Lewis acid-base reaction between SO3 and H2O to form H2SO4 is the reaction that is the primary cause of acid rain. The oxygen atom of the water molecule contains two lone pairs, so water is a Lewis base, while the sulfur atom in SO3 has only three electron regions, which makes SO3 Lewis acidic.What is co32?
CO3 2- is carbonate. a carbonate is a salt of carbonic acid (H2CO3),characterized by the presence of the carbonate ion, a polyatomic ion with the formula of CO3 2-. CO32- is an anion (a negative ion) seen frequently in chemistry. In the CO32- Lewis structure carbon is the least electronnegative element.Is so3 trigonal planar?
The molecular geometry of SO3 is trigonal planar with symmetric charge distribution on the central atom.What is the shape of co2?
Carbon dioxide has two electron groups and no lone pairs. Carbon dioxide is therefore linear in electron-group geometry and in molecular geometry. The shape of CO2 is linear because there are no lone pairs affecting the orientation of the molecule.Which has a trigonal pyramid structure?
Trigonal pyramidal is a molecular shape that results when there are three bonds and one lone pair on the central atom in the molecule. Molecules with an tetrahedral electron pair geometries have sp3 hybridization at the central atom. Ammonia (NH3) is a trigonal pyramidal molecule.How do you find the bond order?
If there are more than two atoms in the molecule, follow these steps to determine the bond order:- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.



