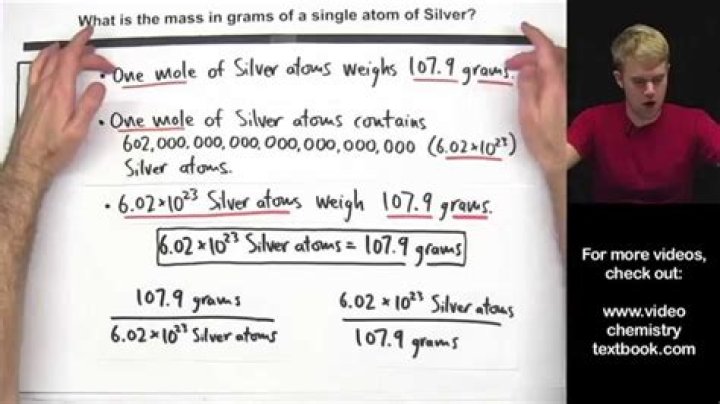It means: The mass of monoatomic element ingrams which will contain 1 mole of its atoms.It is equal to the atomic weight of the element but justwritten with grams suffix. For e.g. Silver hasatomic weight or atomic mass of 107.8682 , so itsgram atomic mass is 107.8682 gm..
Also to know is, what is the mass in grams of one silver atom?
The molar mass of silver is 107.9g/mol.(rounding to 108). As 1 mole = 6.023 x 10^23 molecules(Avogadro's number), 108 g of Silver contains 6.023 x10^23 molecules. So, mass of 1 atom of silver= 108 / 6.023 x 10^23 = 1.7931 x 10^-22 g.
Similarly, what is the mass in grams of one sodium atom? This means that a single atom of sodiumweighs 23 atomic mass units (AMUs), but it also means thatone mole of sodium atoms weighs 23 grams.Hence 6.02 x 10^23 atoms of sodium weight a total of23 grams. This means that if we divide 23 grams bythe number of atoms, we should have the weight of asingle atom.
Similarly, what is the mass of silver atom?
107.8682 u
How do you figure out the mass of an atom?
For any given isotope, the sum of the numbers of protonsand neutrons in the nucleus is called the mass number. Thisis because each proton and each neutron weigh one atomicmass unit (amu). By adding together the number of protons andneutrons and multiplying by 1 amu, you can calculate themass of the atom.
Related Question Answers
How do I calculate moles?
Use the molecular formula to find the molar mass;to obtain the number of moles, divide the mass of compoundby the molar mass of the compound expressed in grams.How do you find the mass in grams of an atom?
To calculate the number of atoms in asample, divide its weight in grams by the amu atomicmass from the periodic table, then multiply the result byAvogadro's number: 6.02 x 10^23.What is the mass in grams of 1.14 mol of water h2o?
The average mass of one H2O molecule is18.02 amu. The number of atoms is an exact number, the number ofmole is an exact number; they do not affect the number ofsignificant figures. The average mass of one mole ofH2O is 18.02 grams. This is stated: the molarmass of water is 18.02 g/mol.How many molecules are in a mole?
Chemists generally use the mole as the unit forthe number of atoms or molecules of a material. Onemole (abbreviated mol) is equal to6.022×1023 molecular entities (Avogadro's number),and each element has a different molar mass depending on the weightof 6.022×1023 of its atoms (1mole).What is the mass of 1 molecule of CO?
The molecular mass of CO is 12.01 + 16.00or 28.01 g/mol. Another common compound with the same elements isCO2 (carbon dioxide). The difference is thatcarbon dioxide has two oxygen atoms instead of justone.What is the mass of 1 mole of nitrogen atoms?
1 mole of nitrogen atoms is equivalent to thegram atomic mass of nitrogen. Therefore, 1 mole ofnitrogen atoms contains 14 g.What is the mass of one atom of carbon?
The mass of a single carbon atom is 1.994 x10-23 g.Who discovered silver?
Silver was one of the first metalsdiscovered by ancient peoples. Silver artifacts havebeen found in many ancient civilizations such as the Sumer from3000 BC. It comes from the Anglo-Saxon word "seolfor" for theelement.How many electrons are there in silver?
47 electrons
How do you find the atomic mass of silver?
Look up the mass of each isotope. The first is the atomic mass, or the massof one atom of each isotope. Isotopes with more neutronshave more mass. For example, the silver isotopeAg-107 has an atomic mass of 106.90509 amu (atomicmass units). The isotope Ag-109 is slightly heavier with amass of 108.90470.What is the mass in grams of a single water molecule?
Step #1 - the molar mass of water is18.015 g/mol. This was calculated by multiplying the atomicweight of hydrogen (1.008) by two and adding the result to theweight for one oxygen (15.999). Please remember that youneed the molar mass first when trying to find themass of one molecule.Where is silver found?
It also is mined from the minerals acanthite(silver sulfide) and stephanite. Silver also isfound in the common minerals chlorargyrite (silverchloride) and polybasite. Silver is mined in many countries,but most comes from the USA, Canada, Mexico, Peru andBolivia.How many grams are in 1 mole of oxygen?
15.9994
How much is a mole of water?
Thus, for example, one mole of water contains6.02214076×1023 molecules, whose total mass isabout 18.015 grams – and the mean mass of one molecule ofwater is about 18.015 daltons. The mole is widelyused in chemistry as a convenient way to express amounts ofreactants and products of chemical reactions.What is the average mass in grams of one uranium atom U )?
One uranium atom has a mass of3.95x10−22 grams.What is the average mass of a single chlorine atom in grams?
If you use 35.45 from the periodic table for theaverage atomic weight of chlorine, 35.45/(6.02*10^23)= 5.88 x 10^-23 gm is your answer.What is the average mass of a single silicon atom in grams?
This is an average for all silicon atomsfrom all the silicon isotopes. As we do not know the isotopeof the atom in question, we'll use the average massfrom the periodic table of elements. Next, we convert the atomicmass units to grams. The mass of an AMU ingrams is 1.661 X 10-24 grams.How do you convert from moles to grams?
Moles to Grams Conversion Formula. In order toconvert the moles of a substance to grams, youwill need to multiply the mole value of the substance by itsmolar mass. More commonly written for this application as: where,is the molar mass of the substance.