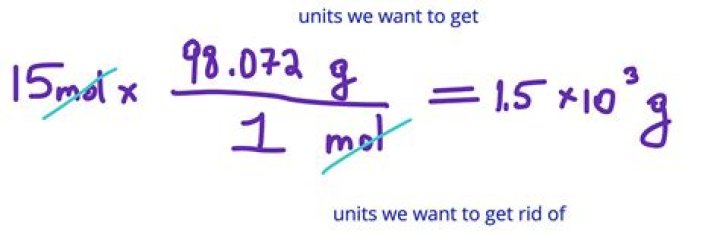If by formula unit you mean the smallest possible unit of Fe2O3, then you are talking about one molecule of Fe2O3. Since the formula weight is grams per mole and there are 6.022 x 10^23 molecules in a mole, you would divide the molar mass by 6.022 x 10^23 to get the mass in grams per molecule..
Accordingly, what is the mass in grams of one molecule?
The average mass of one H2O molecule is 18.02 amu. The number of atoms is an exact number, the number of mole is an exact number; they do not affect the number of significant figures. The average mass of one mole of H2O is 18.02 grams. This is stated: the molar mass of water is 18.02 g/mol.
One may also ask, how do you calculate Mol? Use the molecular formula to find the molar mass; to obtain the number of moles, divide the mass of compound by the molar mass of the compound expressed in grams.
Beside this, what is the mass of 1 molecule?
This can also be written as 6.022×1023 mol-1. The mass of one mole of a substance is equal to that substance's molecular weight. For example, the mean molecular weight of water is 18.015 atomic mass units (amu), so one mole of water weight 18.015 grams.
How many moles are in 36 g of h2o?
Answer and Explanation: There are 4 moles of hydrogen atoms in 36 grams of water, which is choice a.
Related Question Answers
How many grams are in 1 mole of oxygen?
15.9994
How many moles are in o2?
0.031251171918947 mole
What is the mass in grams of one molecule of dimethylnitrosamine?
a. What is the molar mass of dimethylnitrosamine? The molecular formula for dimethylnitrosamine is C 2 H 6 N 2 O. Molar mass = (2 x 12.01) + (6 x 1.008) + (2 x 14.01) + (1 x 16.00) = 74.088 g/mol b.What is the mass in grams of one 12c atom?
The mass of a single carbon atom is 1.994 x 10-23 g.How do you find how many grams are in a mole?
To convert grams to moles, start by multiplying the number of atoms by the atomic weight for each element in the compound. Then, add all of your answers together to find the molar mass of the compound. Finally, divide the number of grams of the compound by the molar mass of the compound to find the number of moles.What is Formula Mass in chemistry?
The formula mass of a molecule (also known as formula weight) is the sum of the atomic weights of the atoms in the empirical formula of the compound. Formula weight is given in atomic mass units (amu).How do you convert mass to Grams?
To convert from g into units in the left column divide by the value in the right column or, multiply by the reciprocal, 1/x. To convert among any units in the left column, say from A to B, you can multiply by the factor for A to convert A into grams then divide by the factor for B to convert out of grams.How many grams are in a atom?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro's number: 6.02 x 10^23.How do you find the mass number of an element?
Together, the number of protons and the number of neutrons determine an element's mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.How do you find the molar mass of an unknown?
Sample Problem: Molar Mass from Freezing Point Depression Step 1: List the known quantities and plan the problem. to calculate the molality of the solution. Then use the molality equation to calculate the moles of solute. Then divide the grams of solute by the moles to determine the molar mass.What is the mass of 1 n2?
Nitrogen gas is defined as N2. its molecular mass will become 28 . According to the mole concept in one mole there is 6.022×10^23 moles are present.so mass of one molecule of nitrogen gas 28/6.022×10^23.What is mass of 1 mole of water?
18.01528 g/mol
What is the standard unit of mass?
kilogram