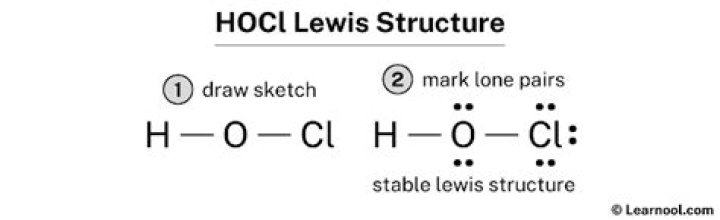
For the HOCl Lewis structure, calculate the total number of valence electrons for the HOCl molecule. After determining how many valence electrons there are in HOCl, place them around the central atom to complete the octets. There are a total of 14 valence electrons in the Lewis structure for HOCl..
Keeping this in consideration, what is the Lewis structure for HCLO?
The Lewis structure of hypochlorous acid has oxygen (O) with single bonds between hydrogen and chlorine. In the Lewis structure, we see that hypochlorous acid has 14 valence electrons. Four are used as bonding electrons, and the remaining 10 are nonbonding electrons on oxygen and chlorine.
Secondly, how many lone pairs are in HOCl? HOCl has H single bonded to O single bonded to Cl. O has 2 lone pairs and Cl has 3 lone pairs. 14 electrons total.
Herein, what is the shape of HOCl?
Hocl molecular shape - You can only upload photos smaller than MB. Because there are only two atoms geometry that makes sense is Linear. Two of the orbitals are filled oxygen has six valence electrons so with and have single.
How do you make HOCl?
The disclosed methods of making HOCl involve mixing together in water in an air-free environment, a compound that generates a proton (H′) in water and a compound that generates a hypochlorite anion (OCl—) in water to thereby produce air-free hypochlorous acid.
Related Question Answers
What is the Lewis structure for CoCl2?
Valence Electron Number For CoCl2 (Phosgene gas): C = 4; O = 6; Cl = 7. The molecule is not ionized and has a neutral charge. Therefore, the total amount of valence electrons is 4 + 6 + (7x2) = 24.How do you draw Lewis structures?
These instructions outline the Kelter strategy to draw Lewis structures for molecules. - Step 1: Find the Total Number of Valence Electrons.
- Step 2: Find the Number of Electrons Needed to Make the Atoms "Happy"
- Step 3: Determine the number of bonds in the molecule.
- Step 4: Choose a Central Atom.
What is the Lewis structure of ammonia?
The Lewis structure of ammonia, NH3 , would be three hydrogen atoms bonded to a nitrogen atom in the middle, with a lone pair of electrons on top of the atom. This is the reason why ammonia acts as a Lewis base, as it can donate those electrons.How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.Is BeCl2 tetrahedral?
Since two sp hybridized orbitals form the bond angle of 180°, BeCl2 is necessarily linear. The bond angle made by two sp3 hybridized orbitals is 109.5° (tetrahedral angle).Is so3 2 a planar?
There should be 1 double bond, 2 negatively charged oxygens, 1 neutral oxygen, and 1 neutral sulfur there. And now to answer your question: to use VSEPR theory to determine that its molecular geometry is trigonal planar, you first have to know what VSEPR (Valence Shell Electron Pair Repulsion) theory can be all about.Is HOCl polar or nonpolar?
Hypochlorous acid is HOCl. Here Oxygen atom is sp3 hybridised. Hence, it has bent shape around oxygen owing to presence of two lone pairs. This causes net Dipole moment (0.37 D) and hence it is a polar molecule.What is the valence of hydrogen?
Hydrogen's valence number is one, because it has only one valence electron and needs only one shared electron to fill its energy levels. This means it can bond with many elements. For example, four hydrogen atoms can bond with a carbon atom, which has four valence electrons, to form methane.Is BeCl2 polar or nonpolar?
If the pull from each of the bonds cancels each other out, like forces that are equal in magnitude but opposite in direction, the molecule will be nonpolar. Examples of this are BeCl2, AlCl3 and CF4. Although the bonds are polar, the entire molecule does not have one side that is more or less negative than another.Is h2o2 polar or nonpolar?
Hydrogen peroxide has two equivalent central atoms with two unshared pairs and two single bonds. This creates an angled bond, creating the bent shape of the molecule. Also, because the atoms do not all lie in the same plane, the dipoles do not cancel eachother out. So, hydrogen peroxide is a polar molecule..What is the molecular geometry of becl2?
linear
Is so3 linear?
Sulfur trioxide has trigonal planar molecular geometry since all three S=O. bonds are nearly double bonds and all are the same length, and energy, and are 120 degree apart. Using VSEPR theory we use the sigma bonds to establish the geometry: three bonding pairs and no lone pairs on the central atom = trigonal planar.What is HOCl used for?
The germicidal properties of HOCl have been well reported. Hypochlorous acid is widely used as a disinfectant, for example, in sanitizing wash solutions and swimming pools. In these applications, the reactive chemical is formed in solution by the addition of chlorine to water.Is hypochlorous safe?
You might be surprised to learn that your immune system's fighter, hypochlorous acid, is actually a type of chlorine. It's so gentle that it's FDA approved for use in wound care and eye care products and is also common in veterinary care.Is hypochlorous acid safe for eyes?
Avenova is the only pure hypochlorous acid for eyes (HClO) product shown to safely manage certain eye health issues and conditions. Avenova is the only hypochlorous acid product that is FDA cleared for safe, long-term use while reducing bacterial load on the lids and lashes, helping to keep your lids & eyes healthy.Is hypochlorous acid dangerous?
HOCl is one of the only agents that is both nontoxic to the delicate cells that can heal our wounds while being lethal to almost all known dangerous bacteria and viruses that threaten our health.Is hypochlorous acid strong or weak?
Chemical properties: HOCl is a strong oxidizer and can form explosive mixtures. In aqueous solutions, being a weak acid, it partially dissociates into the hypochlorite ion (OCl-) and H+. HOCl reacts with bases to form salts called hypochlorites.Does hypochlorous acid bleach clothes?
Hypochlorous Scent and Questions About Bleach: Hypochlorous has a unique faint smell that is undetectable to some users, while some equate it to a spa or the ocean, while some confuse it with a faint bleach smell. Hypochlorous (HOCL) is not bleach. Laundry bleach is actually Sodium Hypochlorite (naCIO).