What is the Lewis structure for c3h4?
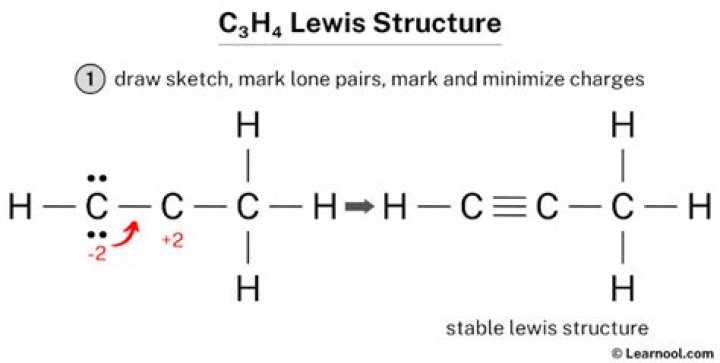
.
Besides, what is the shape of c3h4?
The bond angle formed by the three carbon atoms is 180°, indicating linear geometry for the central carbon atom. The two terminal carbon atoms are planar, and these planes are twisted 90° from each other.
Subsequently, question is, how many double bonds does c3h4 have? two double bonds
Likewise, people ask, what is the Lewis structure for c3h6?
This is the C3H6 Lewis structure. For C3H6 we have a total of 18 valence electrons. The thing about C3H6, is there's more than one way to draw it based on the chemical formula that we're given here. So let's look at the two ways you can draw the C3H6 Lewis structure.
What is the Lewis structure for c2h4?
Drawing the Lewis Structure for C2H For C2H4 you have a total of 12 total valence electrons. Drawing the Lewis structure for C2H4 (named ethene) requires the use of a double bond. In a double bond two pairs of valence electrons are shared (for a total of four valence electrons).
Related Question AnswersWhat shape is c4h10?
tetrahedralWhat shape is c2h4?
trigonal planarWhat is the hybridization of c3h4?
In allene two types of C are present centeral C is sp hybridized while both terminals are sp2 hybridized.Why is c3h4 not planar?
Why is Allene not planar? - Quora. Because of steric hindrances of the overlapping unoccupied p-orbitalls of the double bonded carbon atoms The other side the carbon chain can't have parallel Hydrogens because it will cause greater steric strain.What are the 5 basic shapes of molecules?
Molecular Geometries. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
How many lone pairs are in ch4?
If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3). If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).What is the shape of c3h8?
C3H8 is a linear molecule because it has no specific central atom. Since the molecule is linear, it has 180 degree bond angles.What is the Lewis structure for c2h2?
Drawing the Lewis Structure for C2H2 (Ethyne or Acetylene) For C2H2 you have a total of 10 valence electrons to work with. In drawing the Lewis structure for C2H2 (also called ethyne) you'll find that you don't have enough valence electrons available to satisfy the octet for each element (if you use only single bonds).What c3h6 called?
C3H6 may refer to: Compounds sharing the molecular formula: Cyclopropane. Propene, also known as propylene.How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.How do you draw Lewis structures?
These instructions outline the Kelter strategy to draw Lewis structures for molecules.- Step 1: Find the Total Number of Valence Electrons.
- Step 2: Find the Number of Electrons Needed to Make the Atoms "Happy"
- Step 3: Determine the number of bonds in the molecule.
- Step 4: Choose a Central Atom.



