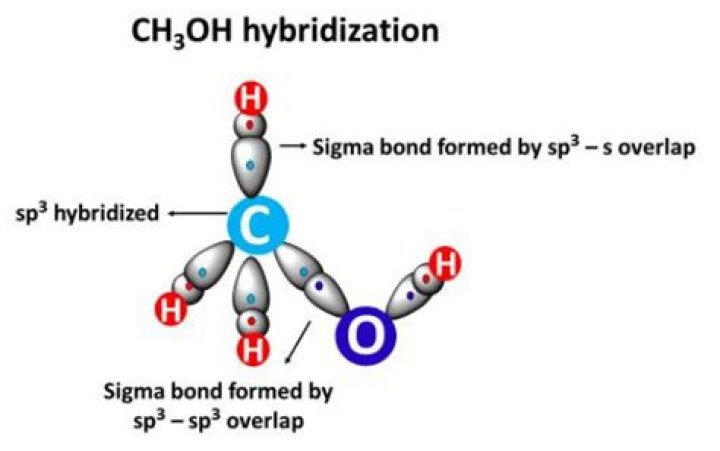
Methanol. The oxygen is sp3 hybridized which means that it has four sp3 hybrid orbitals. One of the sp3 hybridized orbitals overlap with s orbitals from a hydrogen to form the O-H signma bonds..
Hereof, is oxygen a sp2 or sp3?
Answer: The oxygen atom must have either sp2 or sp hybridization, because it needs a p orbital to participate in the C–O π bond. This oxygen atom has three attachments (the carbon and two lone pairs), so we use sp2 hybridization.
Likewise, what is the hybridization of the O atom in formaldehyde? Explanation of Solution Both molecular and electron pair geometries of formaldehyde are trigonal planar. The carbon atom of formaldehyde has sp2 hybridization where one s and two p orbitals mix to form three sp2 hybridized orbital with equal energy. The oxygen atom of formaldehyde has sp2 hybridization.
Secondly, what is the hybridization of HCN?
The carbon atom in HCN is therefore sp hydribidized. The Lewis structure for CH2O shows THREE electron groups surrounding the central carbon atom (two single C-H bonds and one double C-O bond). This carbon atom is sp2 hybridized. So to answer your question, the carbon atom is sp hybridized in HCN and in CO2.
What is the hybridization of the carbon atoms in c2h2?
Answer: Since C2H2 is a linear molecule the C must be sp. Also only sp carbon can form a triple bond. sp2 carbon would give a trigonal planar arrangement. The O in HOCl has two lone pairs and two bonding pairs in a tetrahedral arrangement which is sp3.
Related Question Answers
Why Oxygen has 2 lone pairs?
To give carbon an octet of electrons, we use one of the lone pairs of electrons on oxygen to form a carbon–oxygen double bond: Both the oxygen and the carbon now have an octet of electrons, so this is an acceptable Lewis electron structure. The O has two bonding pairs and two lone pairs, and C has four bonding pairs.Is sp3 an oxygen?
The oxygen is sp3 hybridized which means that it has four sp3 hybrid orbitals. One of the sp3 hybridized orbitals overlap with s orbitals from a hydrogen to form the O-H signma bonds. Due to the sp3 hybridization the oxygen has a tetrahedral geometry.What is the bond order of b2 +?
So the bond order of B2 is equal to 1, which you can get by drawing the molecular orbital diagram and performing the equation Bond Order = . 5 * (# of bonding electrons - # of antibonding electrons). However, when you draw the Lewis structure of B2, you get a triple bond.What does Unhybridized mean?
unhybridized orbitals are in the ground state of the atom. Hybridized orbitals are a combination of orbitals some of which are not in the ground state.Is oxygen in water sp3 hybridized?
The central atom is oxygen, so we do the hybridization with respect to oxygen. Oxygen contains 2 lone pairs of electrons + 2 sigma bonds with the hydrogen atoms, therefore it uses 4 hybrid orbitals. So water shows sp3 hybridization. Oxygen is sp3 hybridised in H2O molecule.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule - Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
Is HCN linear or bent?
Hydrogen Cyanide In this example, HCN, the Lewis diagram shows carbon at the center with no lone electron pairs. The carbon and nitrogen are bonded through a triple bond which counts as "one electron pair". Hence the molecule has two electron pairs and is linear. It boils at 25oC, and thus is a gas a room temperature.Is HCN polar or nonpolar?
Yes HCN is polar. Its structure is H-C≡N. Not only is the nitrogen electronegative intrinsically but it's also tugging on three electron pairs in its triple bond with carbon. In contrast with the single bond with hydrogen on the other side, this makes the molecule polar with a dipole moment towards the N.Is HCN dipole dipole?
(d) HCN is a linear molecule; it does have a permanent dipole moment; it does contain N, however the nitrogen is not directly bonded to a hydrogen. Therefore dispersion forces and dipole-dipole forces act between pairs of HCN molecules.What is the hybridization of c2h4?
When we look at the molecules of C2H4 it has 2 CH molecules and 4 H molecules. The carbon atom consists of 6 electrons and hydrogen has 1electron. The two sp2 hybrid orbitals get overlapped by two hydrogen atoms containing unpaired electrons. A pi bond is formed by the unhybridized 2pz orbitals of each carbon atom.What are Pi and Sigma bonds?
Sigma and pi bonds are chemical covalent bonds. Sigma and pi bonds are formed by the overlap of atomic orbitals. A sigma bond, σ, resembles a similar "s" atomic orbital, and a pi pond, π, has the same orbital symmetry of the p orbital (again, in both cases when viewed down the bond axis).What is a delocalized pi bond?
A pi bond occurs when two sets of electron orbitals, involved in the bond, overlap. A common example occurs in benzene. A delocalized pi-bond means that this pi system can exist in several conformations and thus is not considered to be in either conformation, but all of them at once.Do diatomic molecules hybridize?
Diatomic molecules will always point compatible σ bonding orbital lobes along the internuclear axis, and be able to pair compatible orbitals, so there is no hybridization in molecules like HCl , NO+ , Cl2 , etc.Why do we invoke atomic hybridisation?
Now that carbon has four unpaired electrons it can have four equal energy bonds. The hybridization of orbitals is also greatly favored because hybridized orbitals are lower in energy compared to their separated, unhybridized counterparts. This results in more stable compounds when hybridization occurs.What orbitals hold the lone pairs on the oxygen atoms in co2?
Bonding in Carbon Dioxide Each oxygen makes 1 sigma bond and also needs 2 orbitals for lone pairs of electrons. These must each be 2sp2 hybridized with a remaining 2p orbital. One of the oxygens will have a 2px orbital to combine with the carbon 2px orbital.Do all atoms hybridize?
Hybridization occurs when an atom bonds using electrons from both the s and p orbitals, creating an imbalance in the energy levels of the electrons. To equalize these energy levels, the s and p orbitals involved are combined to create hybrid orbitals.How is c2h2 formed?
In the formation of C2H2, the carbon atom needs extra electrons to form 4 bonds with hydrogen and other carbon atoms. As a result, one 2s2 pair is moved to the empty 2pz orbital. The 2s orbital in each carbon hybridizes with one of the 2p orbitals and forms two sp hybrid orbitals.What is the hybridization of acetylene?
Bonding in acetylene The carbon-carbon triple bond is only 1.20Å long. In the hybrid orbital picture of acetylene, both carbons are sp-hybridized. In an sp-hybridized carbon, the 2s orbital combines with the 2px orbital to form two sp hybrid orbitals that are oriented at an angle of 180°with respect to each other (eg.