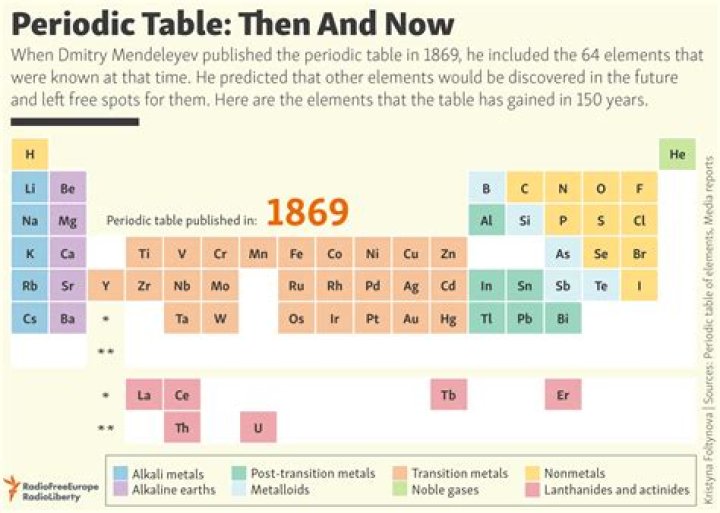
In 1869 Russian chemist Dimitri Mendeleev started the development of the periodic table, arranging chemical elements by atomic mass. He predicted the discovery of other elements, and left spaces open in his periodic table for them. In 1886 French physicist Antoine Bequerel first discovered radioactivity..
Similarly one may ask, how was the first periodic table arranged?
Mendeleev realized that the physical and chemical properties of elements were related to their atomic mass in a 'periodic' way, and arranged them so that groups of elements with similar properties fell into vertical columns in his table.
Beside above, why was the periodic table created? Russian chemist Dmitri Mendeleev published the first recognizable periodic table in 1869, developed mainly to illustrate periodic trends of the then-known elements. He also predicted some properties of unidentified elements that were expected to fill gaps within the table. Most of his forecasts proved to be correct.
Additionally, who invented the periodic table?
Dmitri Mendeleev
How was the periodic table arranged before Mendeleev?
John Newlands. Just four years before Mendeleev announced his periodic table, Newlands noticed that there were similarities between elements with atomic weights that differed by seven. He called this The Law of Octaves, drawing a comparison with the octaves of music. The noble gases (Helium, Neon, Argon etc.)
Related Question Answers
Who invented the periodic table and why?
Dimitri Mendeleev
How many groups are in the periodic table?
18
Who is the father of new periodic table?
Dmitri Mendeleev is often referred to as the Father of the Periodic Table. He called his table or matrix, "the Periodic System".How are elements discovered?
Key Takeaways: How New Elements Are Discovered Superheavy elements are made by striking pre-existing elements with protons, neutrons, or other atomic nuclei. The processes of transmutation and fusion are used.When was the periodic table last updated?
Naming new element On 8 June 2016, IUPAC released the provisional names for the latest 4 elements 113, 115, 117, and 118 – see release and on 28 November 2016, IUPAC announced the approved names and symbols – see release.What is atomic mass in science?
Atomic mass or weight is the average mass of the protons, neutrons, and electrons in an element's atoms.What is the purpose of the periodic table?
What Is the Purpose of the Periodic Table? The periodic table was built to show the relationships among the various elements. The periodic table was constructed in 1869 by Dmitri Mendeleev. Elements are placed on the periodic table based on their atomic structure.Who modernized the periodic table?
Hubbard
What did Moseley do?
Physicist Henry Moseley discovered the atomic number of each element using x-rays, which led to more accurate organization of the periodic table. We will cover his life and discovery of the relationship between atomic number and x-ray frequency, known as Moseley's Law.What is the periodic?
The periodic table, also known as the periodic table of elements, is a tabular display of the chemical elements, which are arranged by atomic number, electron configuration, and recurring chemical properties. The columns, called groups, contain elements with similar chemical behaviours.Who discovered elements?
The first modern list of chemical elements was given in Antoine Lavoisier's 1789 Elements of Chemistry, which contained thirty-three elements, including light and caloric. By 1818, Jöns Jakob Berzelius had determined atomic weights for forty-five of the forty-nine then-accepted elements.What is an element science?
An element is a substance that is made entirely from one type of atom. For example, the element hydrogen is made from atoms containing a single proton and a single electron. If you change the number of protons an atom has, you change the type of element it is. Currently, scientists know of 118 different elements.Which 4 elements are named after planets?
Four of the elements are named after planets (Earth – in the form of tellurium, Mercury, Neptune and Uranus). A further two are named after dwarf plants (Pluto and Ceres), while one after a star (helium from the Greek for the sun – Helios) and another after an asteroid (Pallas) feature on the periodic table.What is a period number on the periodic table?
Periods in the periodic table. In each period (horizontal row), the atomic numbers increase from left to right. The periods are numbered 1 through 7 on the left-hand side of the table. Elements that are in the same period have chemical properties that are not all that similar.Why do we need periodic table?
Before all the naturally occurring elements were discovered, the periodic table was used to predict the chemical and physical properties of elements in the gaps on the table. The table is useful for modern students and scientists because it helps predict the types of chemical reactions that are likely for an element.What is the oldest element in the world?
The oldest chemical element is Phosphorus and the newest element is Hassium.How the periodic table changed the world?
The first periodic table in the "rows and columns" form we see today was invented by Dmitri Mendeleev in 1869. It included the properties of all of the known elements of that time. The periodic table has long-since filled in Mendeleev's gaps and has added new elements. It has even changed the weights of other elements.How many elements were in the first periodic table?
60 elements
Why did scientists not accept Mendeleev's periodic table?
Because the properties repeated themselves regularly, or periodically, on his chart, the system became known as the periodic table. In devising his table, Mendeleev did not conform completely to the order of atomic mass. He swapped some elements around.