What is the difference between an electronegativity and the ionization potential? Electronegativity is the tendency of an atom to attract an electron and make it a part of its orbital. Ionization enthalpy, is the energy required to remove an electron from an atom..
Likewise, how are ionization energy and electronegativity related?
Ionization energy is the energy required to remove an atom's electrons. Electron affinity is the change in energy when an electron is added to form a negative ion. Electronegativity is an atom's attraction to its own electrons and other electrons.
Similarly, why does fluorine have higher ionization energy than iodine? Fluorine has a higher ionization energy because it is in a lower energy level and does not receive the same intensity of shielding as Iodine. Thus Fluorine has a stronger nuclear force on the surrounding electrons and require more energy to remove electrons.
Similarly one may ask, what is the difference between electronegativity and ionization energy quizlet?
(First ionization energy is the energy needed to remove the most loosely held electron from the atom). 6. Electronegativity is the tendency of the atoms of an element to attract electrons when they are chemically combined with another element.
How are ionization energy and electronegativity related to atomic radius?
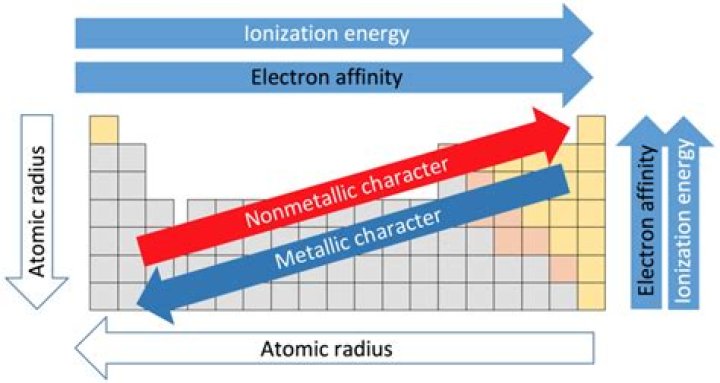
Trends in Atomic Radius, Ionization Energy and Electronegativity. (As the number of protons in the nucleus increases, the nucleus is more attractive to electrons.) Shielding means “blocking”. If electrons are shielded or “blocked” they won't feel the attractive pull of the nucleus.
Related Question Answers
Which element has the highest ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).Which element has the highest electronegativity and ionization energy?
Thus, fluorine is the most electronegative element, while francium is one of the least electronegative. (Helium, neon, and argon are not listed in the Pauling electronegativity scale, although in the Allred-Rochow scale, helium has the highest electronegativity.)What is the general trend in first ionization energy and electronegativity?
Which general trends in first ionization energy and electronegativity values are demonstrated by group 15 elements as they are considered in order from top to bottom? The first ionization energy decreases and the electronegativity decreases.What is meant by first ionization energy?
Definition. The first ionisation energy is the energy required to remove one mole of the most loosely held electrons from one mole of gaseous atoms to produce 1 mole of gaseous ions each with a charge of 1+.Why does electronegativity increase?
Electronegativity increases as you move across the periodic table from left to right. This occurs due to a greater charge on the nucleus, causing the electron bonding pairs to be very attracted to atoms placed further right on the periodic table. Fluorine is the most electronegative element.Is electron affinity equal to ionization energy?
Electron affinity is the energy released when one mole of neutral gaseous atoms gains one mole of electrons to form the -1 anion. The ionization energy of the -1 charged anion is the energy which must be added to remove the “extra” electron and return to the neutral atom. These two energies are equal.Why do nonmetals have high ionization energy?
Why do non-metals have high ionization energy? Because non-metals have to gain electrons, so they have more valence electrons than metals. Therefore, it requires more energy to remove them. When a metal loses an electron, a nonmetal gains an electron.What are the trends for electronegativity and ionization energy similar?
Both depend on the attraction of electrons - electronegativity deals with attraction of bonded electrons, ionization energy deals with the attraction of an atom's own valence electrons. More protons and a smaller atomic radius will increase both.What is the periodic trend for first ionization energy?
The first ionization energy varies in a predictable way across the periodic table. The ionization energy decreases from top to bottom in groups, and increases from left to right across a period. Thus, helium has the largest first ionization energy, while francium has one of the lowest.Why does ionization energy increase across a period quizlet?
Why does first ionization energy increase as you move across a period? The atomic radii (size of the atom) becomes smaller as you move across the period because there is a progressively higher nuclear charge. This makes it harder to remove the outermost electron.Why are the trends for electronegativity and ionization energy basically the same?
3) Why are the trends for electronegativity and ionization energy basically the same? They both measure how much an atom likes to have electrons. Electronegativity measures how much an atom likes to pull electrons away from another one. Ionization energy measures how much an atom doesn't want to lose electrons.What is ionization energy quizlet?
Definition of ionization. First ionization energy is the energy required to pull an electron off a neutral Atom. Second ionization energy is the energy required to pull off an electron off of a plus one charge Adam.What does a negative electron affinity mean?
Electron Affinity is the energy associated with the addition of an electon to a gaseous atom. This is because energy is usually released in this process, as opposed to ionization energy, which requires energy. A more negative electron affinity corresponds to a greater attraction for an electron.What is ionization energy What is the difference between first ionization energy and second ionization energy?
The first ionization energy is the energy it takes to remove an electron from a neutral atom. The second ionization energy is the energy it takes to remove an electron from a 1+ ion. (That means that the atom has already lost one electron, you are now removing the second.)What is the periodic trend for first ionization energy quizlet?
Ionization energy of atoms generally increases from left to right across each row of the periodic table. This is because of the increase in nuclear charge. A nucleus containing more protons has a larger total positive charge, which results in a greater attractive force being applied to each electron.What is meant by atomic radius?
The atomic radius of a chemical element is a measure of the size of its atoms, usually the mean or typical distance from the center of the nucleus to the boundary of the surrounding shells of electrons. The value of the radius may depend on the atom's state and context.Does oxygen or fluorine have a higher ionization energy?
As you can see, fluorine is slightly closer by nature to achieving a full octet than oxygen. Therefore, it is more difficult to remove an electron from fluorine than from oxygen, giving it the higher ionization energy (highest one on the periodic table).Why does fluorine have the highest electronegativity?
Electronegativity of Fluorine Fluorine is the most electronegative element because it has 5 electrons in it's 2P shell. The optimal electron configuration of the 2P orbital contains 6 electrons, so since Fluorine is so close to ideal electron configuration, the electrons are held very tightly to the nucleus.Why does ionization energy decrease from top to bottom?
The ionization energy of the elements within a period generally increases from left to right. This is due to valence shell stability. The ionization energy of the elements within a group generally decreases from top to bottom. This is due to electron shielding.