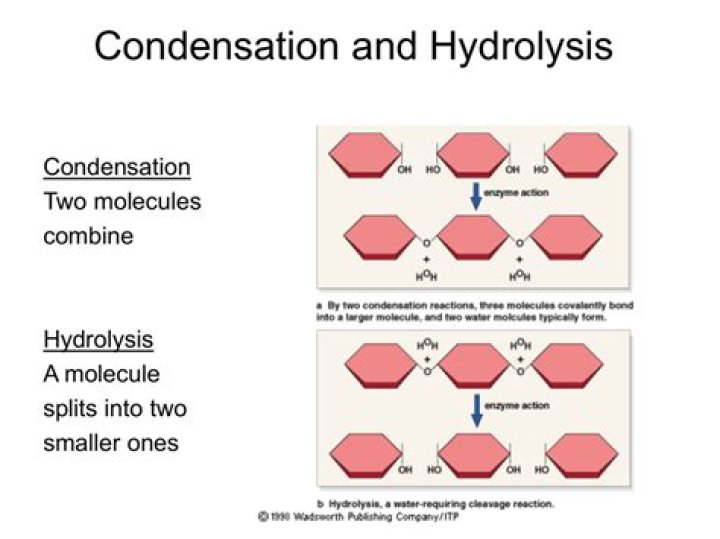
Condensation and hydrolysis are chemical reactions. Hydrolysis always includes water as a reactant. Condensation sometimes give water as a product. The main difference between condensation and hydrolysis is that condensation produces a smaller molecule as a byproduct whereas hydrolysis does not produce a byproduct..
Also know, what are condensation and hydrolysis reactions?
Hydrolysis reactions are the breakdown of molecules after the addition of water while condensation reactions form complex molecules from simpler ones releasing water.
One may also ask, what is a hydrolysis reaction? Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both substance and water molecule to split into two parts. In such reactions, one fragment of the target molecule (or parent molecule) gains a hydrogen ion.
Then, is condensation reaction the same as dehydration synthesis?
Dehydration synthesis is the process of joining two molecules, or compounds, together following the removal of water. During a condensation reaction, two molecules are condensed and water is lost to form a large molecule. This is the same exact process that occurs during a dehydration synthesis.
What is a hydrolysis reaction biology?
Hydrolysis, the opposite of condensation, is a chemical reaction in which water breaks down another compound and changes its makeup. Most instances of organic hydrolysis combine water with neutral molecules, while inorganic hydrolysis pairs water with ionic molecules, such as acids, salts and bases.
Related Question Answers
What is an example of hydrolysis?
Salt. Dissolving a salt of a weak acid or base in water is an example of a hydrolysis reaction. Strong acids may also be hydrolyzed. For example, dissolving sulfuric acid in water yields hydronium and bisulfate.Why are hydrolysis reactions important?
Hydrolysis is an important part of how your body breaks food into its nutritious components. The food you eat enters your body in the form of polymers that are far too large to be used by your cells, so they must be broken down into smaller monomers.What is the opposite of a condensation reaction?
The opposite of a condensation reaction that releases a water molecule is called a hydrolysis reaction. This happens when one molecule is split into two via the addition of a water molecule.What is an example of a condensation reaction?
A simple example is the condensation of two amino acids to form a peptide. This reaction example is the reverse of hydrolysis, which splits a chemical entity into two parts through action from the polar water molecule, which itself splits into hydroxide and hydrogen ions.Why are condensation and hydrolysis reactions important?
Hydrolysis and Condensation reactions are important because they are necessary to the formation and dissociation of the important biological macromolecules. Water can act both as an acid and base. When it acts as an acid, water donates its hydrogen atom. When it acts as a base, water accepts a proton.What is another name for condensation reaction?
Dehydration. Another name for the condensation reaction. Condensation, also called this, refers to the removal of a water molecule during the linking of monomers.Why is condensation reaction important?
The simpler molecule produced is often water, which is why the phrase "condensation reaction" is used, while sometimes being referred to as a dehydration. Condensation reactions are important for the creation of many important biological molecules, such as carbohydrates and proteins.Why is hydrolysis the reverse of condensation?
In fact, Hydrolysis can be the reverse of a condensation reaction in which two molecules join together into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. The carboxylic acids react with the base, converting them to salts.What happens in hydrolysis?
Hydrolysis. Hydrolysis literally means reaction with water. It is a chemical process in which a molecule is cleaved into two parts by the addition of a molecule of water. The most common hydrolysis occurs when a salt of a weak acid or weak base (or both) is dissolved in water.What is an example of a dehydration reaction?
A dehydration reaction is a chemical reaction between two compounds where one of the products is water. Chemicals commonly used as dehydrating agents include concentrated phosphoric acid, concentrated sulfuric acid, hot ceramic and hot aluminum oxide. A dehydration reaction is the same as a dehydration synthesis.What are the products of the hydrolysis reaction?
The reactants other than water, and the products of hydrolysis, may be neutral molecules—as in most hydrolyses involving organic compounds—or ionic molecules, as in hydrolyses of salts, acids, and bases.Is dehydration reaction condensation?
Condensation reaction: Dehydration reaction: In chemistry and the biological sciences, a dehydration reaction is usually defined as a chemical reaction that involves the loss of a water molecule from the reacting molecule. Dehydration reactions are a subset of condensation reactions.Why is it called dehydration synthesis?
This type of reaction is known as dehydration synthesis, which means “to put together while losing water. ” It is also considered to be a condensation reaction since two molecules are condensed into one larger molecule with the loss of a smaller molecule (the water.)What is dehydration synthesis example?
Dehydration synthesis reactions are reactions in which molecules combine by the removal of a H atom and an OH group between them, which together form a molecule of water, H2O . For example, the formation of maltose (malt sugar) from two molecules of glucose is a dehydration synthesis.What occurs during a dehydration synthesis reaction?
Dehydration synthesis is the process of joining two molecules, or compounds, together following the removal of water. During a condensation reaction, two molecules are condensed and water is lost to form a large molecule. This is the same exact process that occurs during a dehydration synthesis.What is the purpose of dehydration reaction?
Dehydration reactions link monomers together into polymers by releasing water, and hydrolysis breaks polymers into monomers using a water molecule. Monomers are just single unit molecules and polymers are chains of monomers.Why are a hydrogen and hydroxyl removed during a dehydration synthesis reaction?
The removal of a hydrogen from one monomer and the removal of a hydroxyl group from the other monomer allows the monomers to share electrons and form a covalent bond. Thus, the monomers that are joined together are being dehydrated to allow for synthesis of a larger molecule.How do you identify a hydrolysis reaction?
Hydrolysis can also be thought of as the exact opposite reaction to condensation, which is the process whereby two molecules combine to form one larger molecule. The end result of this reaction is that the larger molecule ejects a water molecule.What is hydrolysis in the human body?
Hydro- stands for water while -lysis stands for the breaking down of molecules. Hence, hydrolysis is the chemical breakdown of a substance using water. During hydrolysis, a macromolecule is broken down into simpler units. In the human body, all four types of macromolecules are hydrolyzed by water.