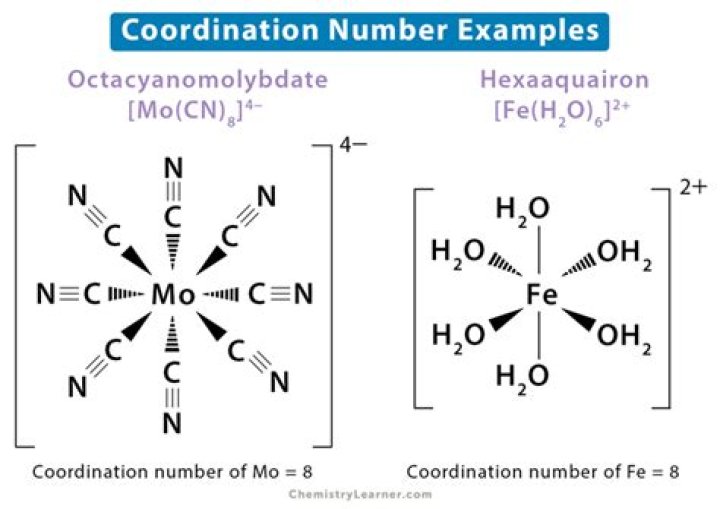
12. What is the coordination number of the Fe atom is K3[Fe(C2O4)3]? Ans. The coordination number is 6..
Furthermore, what is the oxidation state of Fe in k3fe c2o4 3?
+3
Also Know, what is the charge of c2o4? C2O4 is a polyatomic ion with a charge of -2. The oxygen atom in this molecule have a oxidation state of- 2 because oxygen always has -2 charge on it. Since there are 4 atoms of oxygen the overall charge of the oxygen is 4 (-2) =-8. The oxidation states of c is +3 & o is -2.
Besides, what is the coordination number of iron in potassium Trioxalatoferrate 3?
Of ligands attached are 2 therefore coordination no. is 2 “. It is determined the total number of donor atoms in a molecule . Therefore , in the given compound K3[Fe(ox)3] coordination number is 6.
What is the oxidation number of Fe in k4 Fe CN 6?
The oxidation state of iron is +3 in potassium ferricyanide. “Ferri-” comes from “ferric”, the higher of the two oxidation states of iron. Also can be called potassium hexacyanoferrate(III).
Related Question Answers
Is Iron oxalate soluble?
Ferrous oxalate, or iron(II) oxalate, is a inorganic compound with the formula FeC2O4 • xH2O where x is typically 2. These are orange compounds, poorly soluble in water.Why is Ferrioxalate green?
Both are crystalline compounds, lime green in colour. , also known as potassium trisoxalatoferrate or potassium tris(oxalato)ferrate(III). ]3−, which appears fluorescent green in color. The ferrioxalate anion is quite stable in the dark, but is decomposed by light and higher-energy electromagnetic radiation.What is the oxidation number of oxalate?
The oxidation number of carbon atom in oxalate ion changes from +3 to +4. Total change in oxidation number of carbon per oxalate ion is 2. Total change in oxidation number of carbon per molecule of C a ( H C 2 O 4 ) 2 Ca{ left( H{ C }_{ 2 }{ O }_{ 4 } ight) }_{ 2:} Ca(HC2O4) 2 is 4. Thus, the option A is incorrect.Is oxalate a strong ligand?
Oxalate, the conjugate base of oxalic acid, is an excellent ligand for metal ions. It usually binds as a bidentate ligand forming a 5-membered MO2C2 ring. An illustrative complex is potassium ferrioxalate, K3[Fe(C2O4)3].What is the coordination number of CR in k3 Cr Ox 3?
These ligands occupie two positions around the central metal atom. There are 3 oxalate ligands, so it will occupy = (2 × 3) = 6 positions around the central metal atom. Hence, the coordination number of chromium in given complex is 6.How do you find the coordination number?
Here are the steps for identifying the coordination number of a coordination compound. - Identify the central atom in the chemical formula.
- Locate the atom, molecule, or ion nearest the central metal atom.
- Add the number of atoms of the nearest atom/molecule/ions.
- Find the total number of nearest atoms.
What do you mean by coordination number?
Coordination number, also called Ligancy, the number of atoms, ions, or molecules that a central atom or ion holds as its nearest neighbours in a complex or coordination compound or in a crystal.What is c2 o4?
Oxalate(2-) is a dicarboxylic acid dianion obtained by deprotonation of both carboxy groups of oxalic acid. It has a role as a human metabolite and a plant metabolite. It is an oxalate and a dicarboxylic acid dianion.What is c2o4 called?
C2O4−2 is a dicarboxylic acid dianion with chemical name Oxalate. It is also called Ethanedioate or Oxalate Ion or Oxalic Acid Dianion.Is c2o4 a neutral ligand?
Is c2o4 neutral ligand? - Quora. (C2O2−4), is a chelating agent for metal cations. Anhydrous oxalic acid exists as twopolymorphs; in one the hydrogen-bondingresults in a chain-like structure whereas the hydrogen bonding pattern in the other form defines a sheet-like structure.Is c2o4 Bidentate?
Oxalate ion is a bidentate ligand even though it contains four O atoms which have lone pairs of electrons. In this complex, three oxalate ions are bonded to the Fe atom.What is the oxidation number of nitrogen in nh4+?
The oxidation of nitrogen in NH4+ is -3. The hydrogen atoms have +1 and there are four of them. And net charge on the molecule is +1. Let nitrogen's oxidation state be x.What is oxalate ions?
Oxalates are salts of oxalic acid containing oxalate ion. Oxalate ion is a dianion. The calcium metal ion reacts with oxalate ion to form an insoluble precipitate of calcium oxalate, which is the primary constituent of most of the common kind of Kidney stones.How do you get the oxidation number?
The oxidation number of a monatomic ion equals the charge of the ion. The oxidation number of H is +1, but it is -1 in when combined with less electronegative elements. The oxidation number of O in compounds is usually -2, but it is -1 in peroxides. The oxidation number of a Group 1 element in a compound is +1.Is sodium oxalate soluble in water?
About Sodium Oxalate Sodium Oxalateis highly insoluble in water and converts to the oxide when heated (calcined).What is the oxidation state of C in ch5n?
To find the correct oxidation number for C in C2O4 2- (the Oxalate ion), and each element in the ion, we use a few rules and some simple math. When it is bonded to Fluorine (F) it has an oxidation number of +2. Here it is bonded to C so the oxidation number on Oxygen is -2.What is the oxidation number of iron in k3 Fe CN 6?
Let ,in the compound Fe(CN)6^3- ,the oxidation number of Fe is x. now, according to sum rule, x + (-1) × 6 = -3. Thus the oxidation number of Fe in compound is 3.What is k4 Fe CN 6?
Potassium ferrocyanide is an inorganic compound with the chemical formula K4Fe(CN)6. Potassium ferrocyanide is also known as yellow potash prussiate, a yellow crystal.What is the oxidation state of iron na4 Fe CN 6?
the groups ferrocyanide have the charge -4, given from 6 ions CN− with charge -1 and a atom of iron with charge +2. Truong-Son N. Each [Fe(Fe(CN)6)] is then a −1 , so with each [Fe(CN)6]4− being a 4− charge, the bolded iron is a +3 .