What is the Aufbau order?
.
Correspondingly, what does Aufbau mean?
The aufbau principle, from the German Aufbauprinzip (building-up principle), also called the aufbau rule, states that in the ground state of an atom or ion, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels.
what is Aufbau law? The Aufbau principle, simply put, means electrons are added to orbitals as protons are added to an atom. Electrons obey Hund's rule, which states that electrons spread out before they pair up if there are two or more energetically equivalent orbitals (e.g., p, d).
Subsequently, question is, what is the Aufbau principle example?
The Aufbau principle dictates the manner in which electrons are filled in the atomic orbitals of an atom in its ground state. It states that electrons are filled into atomic orbitals in the increasing order of orbital energy level. For example, carbon has 6 electrons and its electronic configuration is 1s22s22p2.
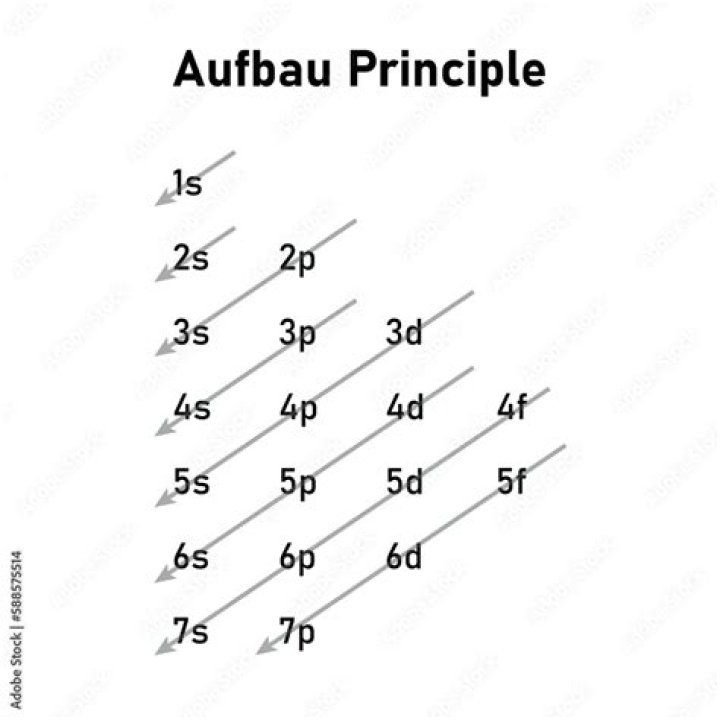
How do you do Aufbau diagram?
The following steps detail how to draw an Aufbau diagram:
- Determine the number of electrons that the atom has.
- Fill the s orbital in the first energy level (the 1s orbital) with the first two electrons.
- Fill the s orbital in the second energy level (the 2s orbital) with the second two electrons.
Who proposed Aufbau principle?
Niels BohrWho gave Aufbau principle?
Niels BohrWho discovered Hund's rule?
Friedrich Hermann HundWhat does Hund's rule of maximum multiplicity States?
The rule states that for a given electron configuration, the lowest energy term is the one with the greatest value of spin multiplicity. This implies that if two or more orbitals of equal energy are available, electrons will occupy them singly before filling them in pairs.Why is the Aufbau principle important?
Aufbau Principle. We can determine the orbitals for the electrons in a multi-electron atoms by placing the electrons into subshells of ever increasing energy. It is important to keep in mind that the Aufbau principle represents and approximate trend that holds in most cases.What does Hund's rule state?
Hund's rule states that: Every orbital in a sublevel is singly occupied before any orbital is doubly occupied. All of the electrons in singly occupied orbitals have the same spin (to maximize total spin).What is Aufbau principle and Hund's rule?
Aufbau Principle: lower energy orbitals fill before higher energy orbitals. Hund's Rule: one electron goes into each until all of them are half full before pairing up. Pauli Exclusion Principle: no two electrons can be identified by the same set of quantum numbers (i.e. must have. different spins).What are the 4 quantum numbers?
Explanation: The four quantum numbers are the principle quantum number, n , the angular momentum quantum number, l , the magnetic quantum number, ml , and the electron spin quantum number, ms .What are the four quantum numbers?
In atoms, there are a total of four quantum numbers: the principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic quantum number (ml), and the electron spin quantum number (ms).Is the Aufbau principle obeyed in all circumstances?
The Aufbau principle holds for almost all elements, especially within the lower atomic numbers. Exceptions are based on the fact that half-full or full shells or subshells are more stable than partially filled ones.What is Hunds rule in chemistry?
Hund's Rule. Hund's rule: every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals have the same spin.What is Hund's rule with example?
Hund's rule : Every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied and all electrons in singly occupied orbitals have the same spin. The image attached is the example of hund's rule.What is SPDF chemistry?
s, p, d, f and so on are the names given to the orbitals that hold the electrons in atoms. These orbitals have different shapes (e.g. electron density distributions in space) and energies (e.g. a hydrogen atom with one electron would be denoted as 1s1 - it has one electron in its 1s orbital.How many energy levels are there?
Number of energy levels in each period The atoms in the first period have electrons in 1 energy level. The atoms in the second period have electrons in 2 energy levels. The atoms in the third period have electrons in 3 energy levels. The atoms in the fourth period have electrons in 4 energy levels.How do you write electronic configuration?
Steps- Find your atom's atomic number.
- Determine the charge of the atom.
- Memorize the basic list of orbitals.
- Understand electron configuration notation.
- Memorize the order of the orbitals.
- Fill in the orbitals according to the number of electrons in your atom.
- Use the periodic table as a visual shortcut.



