What is the activity series for halogens?
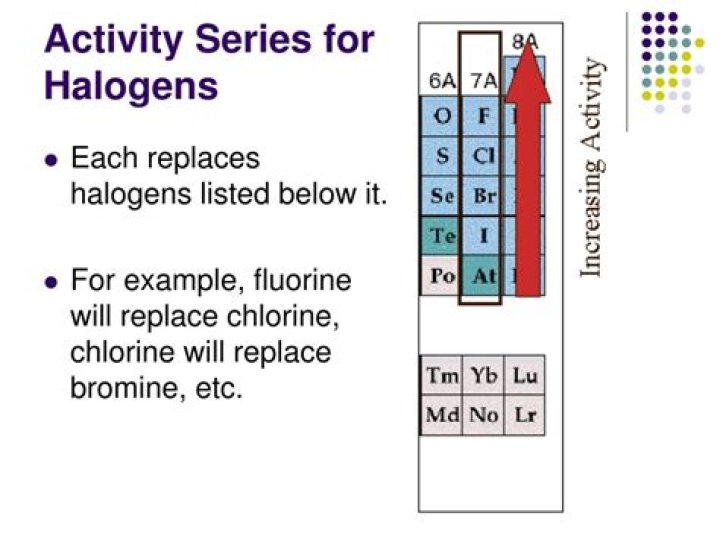
.
Regarding this, what is the reactivity series of halogens?
The order of reactivity is chlorine > bromine > iodine. This is because chlorine could displace bromine and iodine, bromine could only displace iodine, but iodine could not displace chlorine or bromine.
Subsequently, question is, what is the activity series in chemistry? In chemistry, a reactivity series (or activity series) is an empirical, calculated, and structurally analytical progression of a series of metals, arranged by their "reactivity" from highest to lowest.
Then, what is the activity series and how does it work?
The activity series of metals is an empirical tool used to predict products in displacement reactions and reactivity of metals with water and acids in replacement reactions and ore extraction. It can be used to predict the products in similar reactions involving a different metal.
Is there an activity series for nonmetals?
The activity series is a list of elements in decreasing order of their reactivity. Since metals replace other metals, while nonmetals replace other nonmetals, they each have a separate activity series.
Related Question AnswersWhy halogens are so reactive?
Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens can gain an electron by reacting with atoms of other elements. Fluorine is one of the most reactive elements.Which Halogen is most reactive?
fluorineWhy halogens are called halogens?
Group 17 elements are called halogens because halogen is a Greek word which means 'salt producing'. They react with metals to form compounds called salts.Why are the halogens diatomic?
Halogens are diatomic because they have an outer shell of seven valence electrons and react with other atoms of the same element to complete theirWhy is Group 17 so reactive?
Like the alkali metals, the halogens are extremely reactive. They have seven valence electrons, meaning they require only one more electron for a noble configuration. This gives them very large electron affinities and extreme reactivity to form ions with a -1 charge.Why are Group 17 called halogens?
Group 17 elements are called halogens because halogen is a Greek word which means 'salt producing'. Halogens include fluorine, chlorine, bromine, iodine and astatine. They all are non-metals. They react with metals to form compounds called salts.Why does reactivity increase as you go up Group 7?
The reactivity of Group 7 elements decreases down the group. The electrons in the outer shell move further away from the nucleus as we go down the group and the attraction force between the electrons and the nucleus become weaker and weaker. This weaker attraction in the larger atoms makes it harder to gain electron.How many halogens are there?
Types of Halogens Depending on who you ask, there are either 5 or 6 halogens. Fluorine, chlorine, bromine, iodine, and astatine definitely are halogens.Which is the most reactive metal?
cesiumWhy does the activity series work?
Activity Series. The ability of an element to react with another element is called its activity. The easier it is for an element to react with another substance, the greater its activity. For metals, the greater the activity, the easier they lose electrons, forming positive ions.How does the reactivity series work?
The reactivity series is a series of metals, in order of reactivity from highest to lowest. It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series.Is sodium harder than nickel?
1 (c) Sodium (Na) is in Group 1 of the periodic table. Sodium has a higher melting point than nickel. Sodium is more reactive than nickel.Which is more reactive gold or platinum?
It is easier to ionise Platinum so it is more reactive. Though both gold and platinum are among least reactive transition element metals, but still if we look at the reactivity series of metals we find that gold is more reactive than platinum.Is Lithium more reactive than calcium?
don't forget, lithium has one electron in its outer shell while calcium has . these electrons must be lost if the element is to react to form a compound. therefore less power is required to put off the the one electron from lithium than to remove the 2 from calcium, so lithium is more reactive.Why is hydrogen in the activity series?
Hydrogen is a non-metal but it is still included in the reactivity series because it behaves like a metal in quite a lot of chemical reactions. Metals above hydrogen will displace the hydrogen from acids and metals below hydrogen will not displace hydrogen from acids.How do you balance equations?
Method 1 Doing a Traditional Balance- Write down your given equation.
- Write down the number of atoms per element.
- Save hydrogen and oxygen for last, as they are often on both sides.
- Start with single elements.
- Use a coefficient to balance the single carbon atom.
- Balance the hydrogen atoms next.
- Balance the oxygen atoms.