What is reduction reaction explain with example?
.
Besides, what is reduction reaction with example?
Reduction can be considered as the removal of oxygen, the addition of hydrogen, or the gain of electrons. Removal of oxygen: Metal ores which are oxides are reduced to the metal - this is how iron is made from iron ore. The reducing agent is carbon monoxide.
what is an example of oxidation reduction reaction? Another example of a redox reaction is the formation of hydrogen fluoride. We can break the reaction down to analyze the oxidation and reduction of reactants. The hydrogen is oxidized and loses two electrons, so each hydrogen becomes positive. The two electrons are gained by fluorine, which is reduced.
Keeping this in view, what do you mean by reduction reaction?
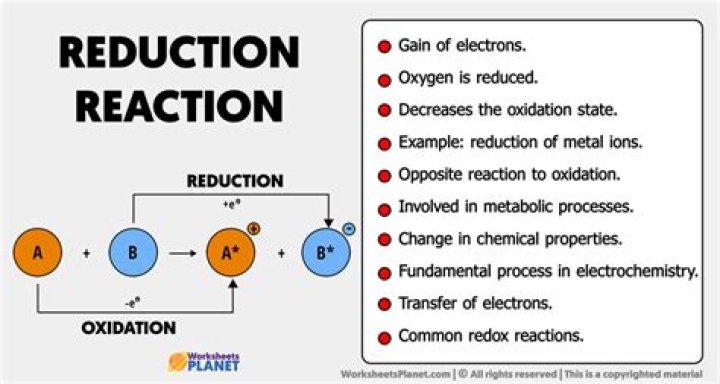
Reduction involves a half-reaction in which a chemical species decreases its oxidation number, usually by gaining electrons. The other half of the reaction involves oxidation, in which electrons are lost. Here, oxidation is the gain of oxygen, while reduction is the loss of oxygen.
What is redox reaction with example Brainly?
Redox reactions are the reactions involving the oxidation of one substance and the reduction of the other. For example - H2s+Cl2-> HCl+S. Hope this helps you!! Answer: Oxidation-reduction reaction or redox reaction is defined as the reaction in which oxidation and reduction reactions occur simultaneously.
Related Question AnswersWhat is the process of reduction?
Reduction is the process of an atom or compound gaining one or more electrons. When an atom or compound gains an electron, its charge gets reduced. The process of reduction is almost always coupled with the process of oxidation. Together, these reactions are called oxidation-reduction reactions, or redox reactions.How do you do Reduction?
Reduction is performed by simmering or boiling a liquid such as stock, fruit or vegetable juices, wine, vinegar, or a sauce until the desired concentration is reached by evaporation. This is done without a lid, enabling the vapor to escape from the mixture.Is Oxygen an oxidizer?
No oxygen is not always an oxidizer or oxidising agent. For it to be so it needs to be reduced, I.e. It needs to gain electrons. For the most part oxygen almost always does this however, if it gets bonded to a much more electro negative atom such as fluorine then oxygen becomes oxidised and the fluorine reduced.What happens in a reduction?
Reduction is the loss of oxygen atom from a molecule or the gaining of one or more electrons. A reduction reaction is seen from the point of view of the molecule being reduced, as when one molecule gets reduced another gets oxidised.What is oxidation reaction with example?
The term oxidation was originally used to describe reactions in which an element combines with oxygen. Example: The reaction between magnesium metal and oxygen to form magnesium oxide involves the oxidation of magnesium. And, each O2 molecule gains four electrons to form a pair of O2- ions.Is respiration a redox reaction?
In summary, cellular respiration is the biological process in which organic compounds are converted into energy. A redox reaction is shorthand for an oxidation-reduction reaction and is a chemical reaction in which one molecule loses electrons while another molecule gains electrons.What is oxidation in simple words?
Oxidation is the loss of electrons during a reaction by a molecule, atom or ion. Oxidation occurs when the oxidation state of a molecule, atom or ion is increased. The opposite process is called reduction, which occurs when there is a gain of electrons or the oxidation state of an atom, molecule, or ion decreases.What is reduction in simple words?
Reduction is a chemical reaction that involves the gaining of electrons by one of the atoms involved in the reaction between two chemicals. The term refers to the element that accepts electrons, as the oxidation state of the element that gains electrons is lowered.What do you mean reduce?
You reduce something when you lessen its volume, size, or degree. Cooks use the word reduce when they reduce a liquid, boiling it down until it has less volume and is thicker.What is oxidation in food?
Oxidation is a reaction that takes place when oxygen has access to products containing fat or pigments. If fats oxidize, they produce “off” odors and flavors (stale, rancid odors). Often, if fats oxidize, vitamins also oxidize and lose their activity.Why is it called oxidation?
In the early days of chemistry, oxidation was defined as a gain of oxygen atoms. the Mg was said to be oxidized because it gained an oxygen atom. Eventually, chemists realized that the reaction involved a transfer of electrons from Mg to O .What is oxidation VS reduction?
Oxidation vs Reduction Oxidation occurs when a reactant loses electrons during the reaction. Reduction occurs when a reactant gains electrons during the reaction. This often occurs when metals are reacted with acid.Is oxidation a rust?
A classic example of a redox reaction is rusting. When rusting happens, oxygen steals electrons from iron. Oxygen gets reduced while iron gets oxidized. The result is a compound called iron oxide, or rust.Is oxidation gaining or losing electrons?
Oxidation is the loss of electrons or an increase in the oxidation state of an atom by another atom, an ion, or a molecule. Reduction is the gain of electrons or a decrease in the oxidation state of an atom by another atom, an ion, or a molecule.What are the two types of oxidation?
Keep this in mind as we look at the five main types of redox reactions: combination, decomposition, displacement, combustion, and disproportion.- Combination. Combination reactions “combine” elements to form a chemical compound.
- Decomposition.
- Displacement.
- Combustion.
- Disproportionation.